Metformin & Weight Loss: Understanding The Link
When someone is given the diagnosis of diabetes, metformin is the first oral drug to be prescribed alongside lifestyle interventions, including changes in diet and physical activity levels. A few people complain of a metallic taste (up to 3%) (Metformin in Noninsulin‐Dependent Diabetes Mellitus) and this should resolve once your body gets used to the medication. It can control blood sugar levels, prevent kidney disease and reduce the risk of heart attack and stroke. Metformin can help with weight loss, particularly for individuals dealing with insulin resistance, PCOS, or type 2 diabetes. Metformin, a medication widely prescribed for managing type 2 diabetes, has been gaining attention for its potential weight loss benefits. This drug may, however, cause weight gain, you could still experience a low blood sugar, and it can interact with other medicines. Though the mechanism by which Metformin causes weight loss is not understood, the drug has been shown in research to help people lose some weight. The drug, when combined with pills that help the pancreas to release insulin, or when combined with insulin, may cause a low blood sugar. Metformin doesn’t increase the pancreas’ release of insulin, so there is a low risk of hypoglycemia, or low blood sugar. Belonging to the class of diabetes drugs, Biguinides, this small pill helps to control blood sugar by lowering it. Understanding how metformin works and its potential side effects empowers patients to take charge of their health. Unlike some other diabetes medications, metformin does not increase insulin production. Metformin is a widely used medication for managing type 2 diabetes, renowned for its ability to lower high blood sugar effectively. The most inspiring “metformin weight loss before and after” stories aren’t just about dropping numbers on a scale; they’re about gaining energy, confidence, and a renewed sense of health. That’s why it’s so important to work with a healthcare provider when considering metformin for weight loss, especially if you’re not diabetic.
Metformin monotherapy appreciably improves dyslipidemia in statin-naive people with T2DM. However, increasing the dosage of metformin yielded no significant effect on its lipid-lowering efficacy. The study enrolled 155 participants with a mean age of 58.6 years and average glycosylated hemoglobin A1c of 8%. Studies have consistently identified dyslipidemia as an important risk factor for the development of macrovascular disease. MedicineNet does not provide medical advice, diagnosis or treatment. When such drugs are withdrawn from a patient receiving Glucophage/Glucophage XR, observe the patient closely for hypoglycemia. Cholestatic, hepatocellular, and mixed hepatocellular liver injury have been reported with postmarketing use of metformin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during post approval use of metformin. Diarrhea led to discontinuation of Glucophage XR in 0.6% of patients.
Community Poll: What Worked Best for Your Diabetes?
These pills contain the drug metformin only. Metformin is an oral medication prescribed for people with Pre-Diabetes and Type 2 Diabetes. If you help to shield them from demise, they will keep your blood sugar down. If you must start taking medication for your newly diagnosed condition, it is then likely that your healthcare provider will prescribe this medication. Regenics offer advanced diagnostic testing to monitor nutrient deficiencies, such as vitamin B12, ensuring that patients can proactively address any gaps in their treatment plans. These symptoms can be particularly noticeable when metformin is first introduced into the treatment plan. Bloating and abdominal discomfort are common gastrointestinal side effects of metformin, often experienced alongside diarrhea or nausea. It typically occurs when the medication is taken on an empty stomach or when the dosage is too high for the body to handle. Regenics is committed to helping patients navigate their health journeys effectively. However, some metformin side effects can be more persistent and may require medical intervention. These typically occur early in treatment and often subside as the body adjusts. The primary mechanism of metformin is its ability to reduce glucose production in the liver, a process known as gluconeogenesis. Instead, it focuses on improving how the body uses existing insulin, making it an essential option for individuals with insulin resistance. In addition to its role in treating diabetes, metformin has been found to have other therapeutic applications. Metformin is an oral medication belonging to the class of drugs called biguanides. The severity can vary greatly among individuals based on dosage, duration of treatment, and personal health factors. Despite its widespread use and efficacy, metformin is not without side effects.
Initial phase II trials demonstrated more weight loss in leflunomide-treated subjects, though in subsequent trials weight loss was not identified as an adverse event (8, 9). Disease modifying treatments (DMARDs) utilized for the treatment of RA might also influence changes in weight. Regression models evaluated changes in BMI with use of each drug compared to methotrexate. This study assessed how primary RA therapies influence changes in body mass index (BMI) in a large administrative database. Unintentional weight loss is important and predicts long-term outcomes in rheumatoid arthritis (RA). The effect of clinically relevant therapeutic dose of metformin on brain bioenergetics and function revealed here may provide insights for the future study of metformin in neurological disorders. Further studies are needed to determine the metformin’s action in middle-age mice, including both male and female, which are more clinically relevant. In the current study, the effect of metformin was only tested in young adult male mice. On the other hand, metformin treatment induced anxiolytic effect and impaired cognitive function upon chronic treatment. A significant increase of AMPK activation was observed in the hippocampus by metformin treatment evidenced by the increase of pAMPK and its downstream TSC2 phosphorylation. Consistent with the decrease of ATP production in vivo, chronic metformin treatment altered major metabolic signals. The reduction of ATP level upon chronic metformin treatment indicated that the action of metformin in glycolytic ATP production is not able to compensate its inhibition of mitochondrial oxidative phosphorylation.
Metformin, a biguanide class medication, has been utilized for decades in the management of type 2 diabetes. Metformin is contraindicated in patients with severe kidney impairment, metabolic acidosis, or known hypersensitivity to metformin. Following the structured Low Carb Program helped Kate lose 19kg and she no longer needs metformin to help manage her diabetes. A number of research studies indicate that metformin may be beneficial in reducing incidence of a variety of cancers. Many people report having to diarrhoea as a result of taking the drug, which in some cases passes over time. The drug helps type 2 diabetics respond better to their own insulin, lower the amount of sugar created by the liver, and decreasing the amount of sugar absorbed by the intestines. This enhances glucose uptake and utilisation by the body. Modified release versions of metformin may be prescribed for people experiencing significant gastro-intestinal intolerance as a result of standard metformin. Metformin is also available as metformin SR, a slow release or modified release form of the medication. Metformin is available both in combination with other drugs, or as a single treatment (a monotherapy). Medication appearance, formulation, and packaging may differ from the images displayed on this website.Prescriptions for any medications require a consultation with a licensed healthcare provider, who will evaluate the patient’s medical condition and determine clinical appropriateness. Viollet et al. (2012) offered a comprehensive overview of the cellular and molecular mechanisms of Metformin in Clinical Science, reinforcing its role in glucose regulation, fat metabolism, and insulin sensitivity. Heck et al. (2000) explored the role of Metformin in managing polycystic ovary syndrome (PCOS), noting its impact on insulin resistance, weight, and hormonal balance in Annals of Pharmacotherapy. Your Alan Health provider can evaluate whether Metformin is appropriate based on your goals, labs (if applicable), and how your body’s responding to treatment. Metformin isn’t a stimulant or appetite suppressant—but it supports weight loss by helping your metabolism work more effectively. Metformin is a well-established medication traditionally used to manage type 2 diabetes. Metformin has been around for decades—but recently, it’s been getting attention for more than just blood sugar control. Also, some participants underwent some changes in their treatment regimen which in turn may have affected the duration of the medication used. Thus, future studies are guaranteed to prospectively measure the outcomes of antipsychotics and metformin concomitant use on AIWG.
Weight loss on metformin can take a few weeks to months. Weight loss is usually a secondary effect, not the main purpose of the medication. Additionally, because Metformin is a prescription medication, it should not be used without medical guidance. That is why it is not recommended for people with certain medical conditions or those who drink large amounts of alcohol. In rare cases, Metformin can lead to a serious condition called lactic acidosis, especially in people with kidney or liver problems. There are also risks, such as vitamin B12 deficiency with long-term use, which need to be monitored. Weight loss with Metformin is often more noticeable when combined with a healthy diet and regular exercise. Others may not lose weight because many factors, including diet and lifestyle, influence weight changes. But they may also play a role in weight management. The answer is not simple because Metformin affects people differently. It is important to remember that Metformin should never be used as a substitute for a healthy diet and exercise. Metformin is a prescription medication, which means it should only be used under the guidance of a doctor. People who drink alcohol heavily should avoid taking Metformin, as it can lead to dangerous side effects. Alcohol can increase the risk of lactic acidosis when combined with Metformin.
This is a dissimilarity of our univariable results which mention that the highest weight gain is in patients who are aged 20–29. Also, in the binary logistic regression model, the highest age group who had weight gain are patients who are 20–29 years old. Gender and weight interval outcomes are similar to the univariable analysis done in this study, which revealed that these two variables have no significant difference in weight. By using a binary logistic regression model with the outcome of decrease/no change in weight and increase in weight, the independent variables of metformin usage, gender, age, and weight interval. However, the mechanism by which these medications induce weight gain is not fully understood (29). However, we estimate that it could be due to comorbidities among elderly patients, namely type 2 diabetes. Little evidence supports our results on the increasing use of metformin among the oldest group of patients (≥70 years). Et al., reported that the amount of weight gain is highest in younger patients, then gradually decreased with the increase in age (5, 24). However, in this study, statistical analysis revealed no significant difference between gender and weight change. We can attribute these differences to the fact that our study was a retrospective cohort study, which justifies, to some extent, the mild inconsistency with other studies. A meta-analysis conducted by de Silva et al. (15) reported similar findings from several clinical studies where metformin effectively reduced the AIWG. AIWG is considered one of the most prominent side effects of these drugs which can lead to the detrimental effects of metabolic syndrome. The other age group categories which are 40–49, 50–59, and 60–69 have no difference regarding weight in comparison to ≥70-year-old patients. Similarly, patients in the 30–39-year-old age group had a 3.58 odds ratio to develop weight gain in comparison to ≥70-year-old patients. In addition, 20–29-year-old patients had a 3.46 odds ratio to develop weight gain in comparison to patients who aged ≥70 years. In the binary logistic regression model, the independent variables were the usage of metformin, gender, age, and weight interval. There was no significant association between group 1 patients and weight interval in qualitative nor quantitative analysis. However, only 3 patients out of 73 (4.11%) in the 20–29 group were reported to use metformin with antipsychotics, the data illustrated in Figure 4. Of note, 18 out of 34 (52.94%) and 12 out of 24 (50%) of patients were reported to use metformin with antipsychotics in the following age groups, 60–69 years and more than 70 years, respectively. Regarding the age variable, a total of 50 out of 73 (68.49%) patients in the 20–29 years age group had an average weight increase of +4.66 kg (95% CI 3.54–5.78).
Drug Interactions
If you are putting on weight on insulin, it will help to speak to your doctor about gradually reducing both your insulin and your dietary intake, particularly with regard to carbohydrate. A problem that can happen for many people who take insulin is that the benchmark of how much needs to be eaten is set by the insulin dose. By working closely with a healthcare provider and making healthy lifestyle choices, women with PCOS and other insulin-resistant conditions can improve their chances of conceiving. Gastrointestinal side effects, such as nausea, vomiting, and diarrhea, are common when first starting metformin. While metformin is generally well-tolerated, it can cause some side effects, including gastrointestinal issues and lactic acidosis. Regular exercise can also improve insulin sensitivity and reduce androgen levels. This may include regular blood glucose monitoring, ovulation tracking, and ultrasound examinations to assess follicular development. Regular monitoring and adjustments to treatment as needed are crucial to maximizing metformin's potential. The optimal dosage and duration of metformin treatment for fertility can vary depending on individual patient characteristics and medical history. At Noom, we know sustainable weight loss isn’t built on hacks, superfoods, or supplements. Lasting weight loss doesn’t come from sprinkling one ingredient into your routine—it comes from stacking habits that work together, day after day. As Dr. Mann summarizes, “Turmeric is not a hack for weight loss, and I wouldn’t recommend it as your sole strategy.” When researchers look specifically at weight loss, turmeric on its own doesn’t deliver meaningful or reliable results. Where things start to unravel is when turmeric gets framed as a weight loss shortcut. There’s no evidence that drinking turmeric in water on an empty stomach boosts fat burning or speeds up weight loss. But meaningful weight loss still comes from consistent habits like balanced eating, regular movement, and behavior change—not from turmeric by itself. Turmeric alone won’t cause weight loss.
The most recent international guidelines recommend that when discussing the use of GLP-1 RA with women with PCOS, the potential side effects and the need for long-term use in weight management should always be considered in the shared decision making with the patient. In addition, basal insulin levels decreased, and insulin resistance (HOMA-IR) improved. There is growing body of evidence that GLP-1 RAs demonstrate numerous metabolic benefits in PCOS, including a high effect on prediabetes remission rate, a significant reduction in atherothrombotic markers, and an improvement in lipid profile (29–32). AOMs, including liraglutide and semaglutide, combined with lifestyle intervention could offer an upgraded treatment strategy for patients with PCOS and obesity (28). Metformin in addition to lifestyle intervention has been used in PCOS over the past few decades (27), and should be considered irrespective of glucose homeostasis in all women with PCOS with BMI ≥ 25 kg/m2, because of its expected beneficial effects on metabolic outcomes (28). Women with PCOS face a 2.8-fold higher risk for obesity and a 1.7-fold higher risk for central obesity compared with individuals without PCOS (25). Improvements of cardiometabolic variables achieved during semaglutide treatment phase reverted to baseline, whereas the reduced free testosterone levels observed during semaglutide treatment did not significantly deteriorate after semaglutide discontinuation. Three subjects experienced intermittent diarrhea, bloating and nausea before the initiation of semaglutide treatment (timepoint 1), but the symptoms were not severe enough to warrant discontinuation of metformin therapy. Two years after semaglutide cessation, 18 women had normal glucose tolerance, 3 had impaired fasting glucose, 3 had impaired glucose tolerance, and 1 developed type 2 diabetes. According to the OGTT at the beginning of the study (timepoint 1), 21 women had normal glucose tolerance (NGT), 3 had impaired fasting glucose (IFG), and 1 had impaired glucose tolerance (IGT). Metabolic improvements during semaglutide treatment phase from timepoint 1 to timepoint 2 included a statistically significant decrease in total and LDL cholesterol, fasting plasma glucose, and glucose on 120 min of OGTT. After discontinuation of semaglutide, they regained about one third of prior weight loss, still resulting in statistically significant net weight loss from the beginning to 2 years after semaglutide cessation. Glucose levels were determined using a standard glucose oxidase method (Beckman Coulter Glucose Analyzer, Beckman Coulter, Inc., CA, USA). A fasting blood sample was drawn for determination of glucose luteinizing hormone (LH), follicle-stimulating hormone (FSH), androstenedione, dehydroepiandrosterone sulphate (DHEAS), total and free testosterone (T), and lipids, followed by a 2-h OGTT. The participants were treated with semaglutide, 1.0 mg/week in addition to their ongoing treatment of metformin at a dosage of 2000 mg/day and lifestyle interventions for 16 weeks.
However, Dr. Saunders says a typical dose can range from 500 to 2,000 mg daily, and you must get a prescription from a doctor to take metformin. “Other potential side effects include nausea, dyspepsia, abdominal pain, gas, headache, dizziness, muscle pain and altered taste.” “Metformin can shift the balance of bacteria in the gut, which plays a role in digestion and possibly in how the body processes and stores energy,” adds Dr. Smith. But between shortages, high out of pocket costs and the need for weekly injections, the medication is not suited for everyone. Secondly, one of the leading causes of hypothyroidism in developed countries is Hashimoto's thyroiditis, an autoimmune condition where the immune system attacks healthy thyroid cells. Firstly, the symptoms of hypothyroidism make it extremely difficult for a person to lose weight. While a slowed metabolism is at the root of weight gain in hypothyroidism, other factors undoubtedly play a role. One of the side effects of a slowed metabolism is difficulty taking energy from stored sources like adipose (fatty) tissue. When there is not enough thyroid hormone available in your blood, it can cause your organs to slow down. What is more, patients with primary hypothyroidism are also three times more likely to develop cardiovascular diseases like atherosclerosis and hypertension earlier in life. Indeed, it is considered safe and well-tolerated only for diabetic patients or those with metabolic syndrome. Hypothyroidism is the condition in which your thyroid hormone production drops causing your body processes to slow down and change. Specifically, it reduces the amount of glucose that you absorb from the food you eat and the amount that is processed and released by the liver. There are some lifestyle changes you can make to help control the symptoms of type 2 diabetes and polycystic ovary syndrome (PCOS). Yes, you can drink alcohol while taking metformin, but it's best to drink no more than 2 units per day.
But have you heard of a medication named Metformin for weight loss in PCOS? Metformin causes weight loss in overweight and obese patients, and also positively affects outcomes in pregnant women. Moreover, a stronger induction of apoptosis was observed in the case of combination therapy compared to the treatment with a single drug . Ulrike Wokoun et al. investigated whether the combination of metformin and 2-deoxy-D-glucose increased antitumor efficacy in in vitro studies. In in vitro studies, the combination of metformin and aspirin significantly inhibited cell migration and pancreatic cancer cell colony formation – PANC-1 and BxPC3 – compared to untreated cells or compared to cells treated with single compounds. It has been shown that the use of metformin in combination with other anti-cancer drugs can potentiate their effects. In 2019, the impact of type II diabetes and certain diabetes on the risk of different molecular subtypes of breast cancer was assessed in a retrospective, multicentre study of over 4500 women with breast cancer . In vitro studies performed in human doxorubicin resistant breast cancer cell line (MCF-7 DX) have shown that metformin administration can reverse multidrug resistance (MDR) by reducing P-gp activity. The results obtained from the analysis did not confirm the association between metformin treatment and incidence of major cancers (excluding prostate and pancreas) in diabetic patients . Dankner et al. conducted a study of 320,000 diabetic patients aged 21–87 years to assess the association between metformin and cancer incidence. A meta-analysis published in 2018, including 20 million people, found that diabetes is a risk factor for cancer of any location in both men and women . One quarter of patients using metformin and 1/8 of patients using placebo had a reduced level of vitamin B12 . These studies included patients of any age or sex, taking any dose of metformin for any period. In 2016, a systematic review and a meta-analysis were performed, which assessed the relationship between the use of metformin and vitamin B12 deficiency in people with type 2 diabetes. The survival rate of female patients using metformin was compared with the survival rate of patients not using metformin. However, children of mothers taking metformin during pregnancy at the age of 9 years had higher values of body weight, arm and waist circumference, and waist circumference-to-height ratio .
We also studied whether weight change from baseline to years 1, 2, and 3 affected the relationship between insulin secretion and sensitivity within each treatment group. To investigate whether the relationship between insulin secretion and sensitivity changed over time, we compared the lines describing this relationship at baseline and at years 1, 2, and 3 separately for each treatment group. Participants were randomized to ILS, metformin, or placebo treatment groups. The DPP was a multicenter, randomized clinical trial that investigated the effects of an ILS and metformin on the prevention or delay of type 2 diabetes onset in participants with IGR (10). We examined the longitudinal associations of weight loss with insulin secretion compensation and secretory demand. These rightward shifts were consistent with increased insulin sensitivity and improved beta-cell function and with the reduced incidence of diabetes for participants in these 2 treatment groups. In healthy individuals, insulin secretion increases to compensate for decreased insulin sensitivity which maintains normal glucose tolerance (4–6). Decreased insulin sensitivity and reduced insulin secretion characterize the transitions from normal glucose regulation (NGR) to impaired glucose regulation (IGR) and from IGR to type 2 diabetes (1–3). ILS and metformin decreased secretory demand while increasing compensatory insulin secretion, with greater effects of ILS. In New York City (NY), where the fast-paced lifestyle and abundance of food options can make weight loss challenging, metformin can be a helpful addition to your weight management strategy. These include high-sugar foods and beverages, as they can cause blood sugar spikes and interfere with the medication's effectiveness. Like any medication, metformin may have side effects, such as gastrointestinal discomfort or vitamin B12 deficiency. While metformin can be a useful tool for weight loss, there are some considerations to keep in mind. If you're considering using metformin for weight loss, it's important to remember that it should be used as part of a comprehensive weight loss management plan. They can also help you develop a plan to transition off the medication if necessary, and provide guidance on maintaining weight loss through sustainable lifestyle changes. Studies have shown that significant weight loss can occur within 6-12 months of starting metformin. Additionally, metformin is a medication that should only be used under the guidance of a healthcare professional, and they will determine if it’s appropriate for your specific situation. It’s crucial to discuss any concerns or side effects with a healthcare professional, as they can adjust the dosage or recommend alternative treatments to minimize side effects. Additionally, regular monitoring and adjustments to the dosage and treatment plan can help maximize weight loss results. A healthy diet and regular exercise are still crucial for achieving and maintaining weight loss.
BMI, body mass index; IFG, impaired fasting glucose; IR, Insulin resistance; PCOS, polycystic ovary syndrome; T2DM, type 2 diabetes mellitus; WMD, weighted mean difference. BMI, body mass index; IFG, impaired fasting glucose; IR, Insulin resistance; MET, metformin; PCOS, polycystic ovary syndrome; RCT, randomized controlled trial; T2DM, type 2 diabetes mellitus. The 21 articles, which focused on the effects of metformin in overweight and obese patients, were published from 2000 to 2018. However, metformin has not been officially approved as a weight-reduction medicine.4 Different reports indicate inconsistencies in the weight loss effects of metformin. But whether this decreased value produced enough weight loss (5% of baseline body weight) to qualify as a “weight loss drug” as current guidelines require, requires larger specific randomized control trials. Metformin aids weight loss by improving insulin sensitivity, reducing glucose production in the liver, and potentially lowering appetite. The average weight loss on metformin varies, but studies suggest that individuals may lose around 5-10 pounds over several months. The truth is that while Metformin can help some people lose weight, its primary function is to manage blood sugar. Some people lose weight on Metformin because it reduces appetite, affects gut bacteria, and helps the body use insulin more efficiently. These actions help people with type 2 diabetes keep their blood sugar under control. People with conditions like diabetes, PCOS, or insulin resistance may see some benefits, but those without these conditions may not experience much weight loss.
Metformin and vascular smooth muscle cells
For people with prediabetes or early type 2 diabetes, berberine might offer meaningful metabolic benefits. It may also interact with blood thinners, blood pressure medications, and certain antibiotics. This timing capitalises on the compound's glucose-lowering effects while potentially reducing digestive side effects. Berberine shares the digestive concerns but lacks metformin's extensive safety monitoring and established protocols for managing side effects. Metformin has decades of safety data and proven cardiovascular benefits beyond blood sugar control. This built-in safety mechanism means people without diabetes are unlikely to experience hypoglycemic episodes. This multi-target approach explains its consistent effects across different studies and populations. Participants experienced reductions in fasting blood glucose by 0.82 mmol/L, HbA1c by 0.63%, and two-hour post-meal glucose by 1.16 mmol/L. Semaglutide (Ozempic's active ingredient) works as a GLP-1 receptor agonist, directly mimicking hormones that regulate blood sugar and slow gastric emptying. Patients who are considering berberine as a supplement should seek medical advice to assess compatibility with their current diabetes medication and to ensure liver function enzymes remain normal. The potential benefits of berberine also extend to its antimicrobial effects, which can aid in preventing bacterial overgrowth in the gut - a condition sometimes exacerbated by diabetes. Patients with Type 2 diabetes, specifically, often face challenges related to insulin resistance and metabolic syndrome. Consulting with a healthcare professional ensures it aligns with your health goals and doesn't interfere with any medications you may be taking. Berberine may assist with weight management due to its ability to improve insulin sensitivity and metabolic rate.
Alcohol use was low in the DPP cohort because heavy drinkers were excluded, and the level of intake (as measured by the Block food frequency questionnaire) remained stable for the duration of the study. Over the study enrollment period of ~3 years, baseline ALT concentrations fell slightly while the 3- and 6-month ALT concentrations rose slightly. In addition, unlike AST in our study, examination of the ALT assay by calendar time did not demonstrate assay drift over the study. First, ALT rose slightly over the study period (Figure 1), but this did not account for the differences seen as study time was included as a covariate in the analysis. That lowering of FI was not more effective in preventing abnormal ALT was surprising, as pioglitazone, an insulin sensitizer, decreases ALT and improves liver histology (7). However, many of the published studies in humans to date did not avoid bias or minimize potential confounders by employing randomization. Metformin also lowers aminotransferase levels and decreases liver fat content in both murine models of NAFLD (21) and humans with NASH (8,22). Serum ALT levels are often used to screen for liver disease, including nonalcoholic fatty liver and NASH. ALT rose over the study period, largely during the first 2 years. Average serum ALT was 17 U/l and levels were above the National Health and Nutrition Examination Survey 95 percentile, sex-specific cut points in 7%. Cox proportional hazards regression was used to examine the effects of changes in all variables at year one with development of abnormal ALT. Cumulative incidence of development of elevated ALT values were analyzed by quartiles of change in weight, FPG, and FI concentrations from baseline to year 1. Examinations were counted whether or not the participant had developed diabetes. To approximate a normal distribution, ALT levels were log transformed. Mean AST concentrations increased in 1999 by ~4 U/l regardless of study visit (baseline, 3, 6 months etc.) consistent with assay drift. Both assays were evaluated for drift over the course of the study.
These recommendations are based primarily on the glucose-lowering effects, relatively low cost, and generally low level of side effects of metformin . Metformin therapy is the initial treatment for patients with Type 2 diabetes according to the current guidelines of the American Diabetes Association/European Association for the Study of Diabetes and the American Association of Clinical Endocrinologists/American College of Endocrinology 1,2. Our results also suggest that metformin treatment may reduce the risk of major coronary events (-4-5%) and all-cause mortality (-2%) in elderly diabetic populations. However, in the elderly an additional unintentional weight loss could be considered as an adverse effect of the treatment. The dosage of metformin and incorporating lifestyle modifications are important factors in the success of this drug working to reduce weight. Yes, they can help with regulating blood sugar and supporting a healthy diet. The recommended dosage for metformin for women with PCOS looking to achieve losing weight may vary depending on individual factors and goals of treatment. An essential factor in the metformin’s body mass index reducing effects of metformin is the dosage. The exact mechanism behind how metformin assists people lose weight for each individual is still unclear. The mechanism of action of metformin involves making the body’s cells more sensitive to insulin, potentially mitigating the insulin resistance common in PCOS sufferers. Elevated glucose levels can lead to glucose intolerance, resulting in weight gain and an imbalance in other hormone levels that may exacerbate the symptoms of PCOS. This article will explore how metformin impacts weight loss in women with PCOS and the research behind these claims. Metformin is used for treating type 2 diabetes mellitus and impacts how your body regulates insulin. While medications to assist with weight loss are available, they are not guaranteed when you join the program. As a result, an individualized weight loss plan is best when you have T2D or prediabetes. Metformin dosage may also affect the degree of weight loss.4,5 During a 16-week study, participants with T2D taking 1000 mg twice daily lost an average of 2.2 pounds (lbs). Some studies indicate metformin has little or no effect on reducing the risk of heart disease, and more research is needed in order to establish metformin's cardiovascular benefits. Metformin is used for the treatment of T2D.1-6 When an individual with T2D is diagnosed, metformin is the first drug prescribed for treatment. This article explains metformin and answers the question, “Does metformin cause weight loss?
Klein et al. reported its efficacy of reducing weight gain in children and adolescents who were treated with olanzapine as well (Klein et al., 2006). Paresthesia was the most common side effect observed in most of the included studies (Ko et al., 2005; Afshar et al., 2009; Narula et al., 2010). In our current review, it showed excellent weight and BMI reduction. Sibutramine is an antiobesity medication affecting both serotonin and norepinephrine reuptake (Henderson et al., 2007). Hence, by using network meta-analysis we were able to allow indirect comparisons between the add-ons as well as against placebo. A similar pattern was seen for BMI change, where all add-ons except cardiac Ranitidine showed statistically significant BMI reductions comparing to placebo. For the BMI change outcome, 24 studies were included in the analysis. However, Reboxetine was reported only in studies with less than 12 months’ follow-up, we were unable to check its sensitivity results. In the sensitivity analysis by excluding the eight studies, similar pooled estimates were obtained and the rank order remained the same. Efficacy results on Topiramate were reported in 4 studies, Metformin in 13 studies, Reboxetine in 3 studies, Ranitidine in 2 studies, and Sibutramine in 4 studies (Figure 1). The latter method first fits an inconsistency model and then conduct a Wald test to check whether there is significant inconsistency among the included studies (White, 2015).
The weight gain between different age groups is found to be elevated among the youngest group (20–29 years) and lowest among the group of 60–69 years old. The elevated proportion of females taking metformin with antipsychotics can be explained by some other indications for metformin use such as polycystic ovary syndrome as one of the obesity’s complications in females (16, 25). These quantitative results are in line with the existing evidence that metformin is effective in controlling AIWG. Gender and weight interval also have no significant difference on weight in binary regression model using the aforementioned variables. The two dependent variables were decrease/no change in weight, and the other variable was increase in weight. The percentage of concomitant metformin use increased throughout the age groups with the youngest having the least frequency and the two oldest age groups (60–69 and ≥70) reported the highest frequencies of 52.94 and 50%, respectively. The youngest two age groups (20–29 and 30–39) showed the highest frequency of weight gain of 68.06 and 70.30%, respectively. No weight changes were most common among the two oldest age groups (60–69 and ≥70) with frequencies of 26.47 and 37.50%, respectively. Importantly, statistical analysis revealed no significant association between age and weight interval. Illustrates the difference between male and female participants in metformin concomitant use.
Primarily being an antihyperglycemic agent, metformin also has a plethora of pleiotropic effects on various systems and processes. Get an online consultation, receive doctor-approved treatment, and have your metformin delivered right to your door for just $30 per month. Some people describe the initial side effects as "terrible diarrhea" or "period-like cramps." Personalized treatment plans that account for your individual needs and health status tend to be more successful. Avoid taking metformin on an empty stomach to minimize side effects. Focus on eating balanced meals with protein, vegetables, and complex carbohydrates to support your weight loss goals. Some people have genetic variations that affect medication metabolism. Especially as the medication can make it easier to stick to a healthy diet by reducing cravings and appetite. Taking your medication at the same time daily helps maintain steady levels in your system. Your baseline metabolic health also influences how well you respond to treatment. Those closer to their ideal weight may experience slower or minimal weight loss results. Metformin dosing for weight loss typically starts low and increases gradually. During your first month, expect side effects like nausea and diarrhea as your body adjusts. Metformin is a first-line medication primarily used to treat type 2 diabetes.
Certain conditions, like weakened heart, kidney, or liver function prohibit the use of metformin even in those with full-fledged diabetes. This general consensus is that metformin must be used in addition to dieting to have its best weight-related effect. Decreasing the amount of food consumed leads to calorie restriction and subsequent loss of body weight. Lifestyle modifications, such as implementing a balanced diet and engaging in regular physical exercise, can enhance the weight-loss effects of Metformin. These symptoms can lead to a reduced appetite or the avoidance of certain foods, which may result in weight loss for some individuals. Metformin can lead to several side effects that may indirectly influence weight. Healthier lifestyle choices, such as a balanced diet and regular exercise, are essential components of any successful weight loss plan. Moreover, it’s important to recognize that Metformin should not be considered a standalone solution for weight loss. Using Metformin specifically for weight loss is a topic of ongoing research and debate. Additionally, Metformin’s ability to influence glucose metabolism plays a significant role in weight management. This may lead to a gradual reduction in body weight, especially when combined with dietary modifications and regular physical activity. By prioritizing healthy eating, regular exercise, and continuous monitoring, many can harness the benefits of Metformin while remaining within their desired weight range. Patients are encouraged to work closely with their healthcare teams to set realistic goals and navigate their weight management journey effectively.
Women with polycystic ovary syndrome (PCOS) may experience weight loss and improvements in insulin resistance after using metformin. People taking these medications found that using metformin helped counteract the weight gain from these medications and support weight loss. Metformin helps control weight gain by preventing high blood sugar levels and reducing appetite. High blood sugar levels can often contribute to weight gain. Incorporating healthy habits, such as consuming a balanced diet rich in whole foods and engaging in regular exercise, can enhance the weight loss effects of Metformin. While Metformin is primarily prescribed for diabetes management, some studies suggest it may aid in weight loss for non-diabetic individuals as well. Whether or not you decide to pursue metformin for weight loss, understanding its mechanisms, potential benefits, and risks can help you make an informed decision. In conclusion, while metformin shows potential for weight loss, especially in insulin-resistant individuals, it’s essential to approach its use with caution. While metformin may assist in weight loss, it is not a substitute for healthy lifestyle changes. Recent research has indicated a connection between metformin usage and weight loss, particularly among individuals with insulin resistance, obesity, or metabolic syndrome. However, its potential benefits extend beyond diabetes control, leading many to wonder if metformin can also aid in weight loss. This may help enhance the weight loss effects of metformin, but always talk to your doctor before adding any new supplements to your daily routine. According to research published in the journal Biology, berberine can help lower blood glucose levels, boost insulin production and reduce insulin resistance. So while metformin may not offer the same dramatic results as weight loss medications like Wegovy, it can be an effective option for long-term weight loss.
Subgroup stratified analysis was conducted to further analyze the effect of metformin on weight loss, as shown in Table 2. The performance of lifestyle interventions during metformin treatment was not mentioned in eight studies23–28,32,36 (Table 1). The present meta-analysis aimed to further investigate the effectiveness of metformin on weight loss, explore the feasibility and effectiveness of metformin as a potential weight loss medicine, and possibly provide a basis for the establishment of clinical obesity management and obesity management guidelines. This meta-analysis aimed to summarize the weight loss effect of metformin quantitatively. However, metformin has not been officially approved as a medicine for weight loss because its effect on different populations remains inconsistent. While no special diet is required, a balanced, low-carb, and high-fiber diet can enhance metformin’s effectiveness for weight loss. Taking metformin without food can cause stomach discomfort, nausea, or even low blood sugar in some individuals. Yes, metformin can reduce appetite in some individuals, which may contribute to weight loss. A 500mg dose of metformin is relatively low and may not be sufficient for significant weight loss. Most people experience modest weight loss, and significant weight reduction usually requires lifestyle changes such as improved diet and increased physical activity. While metformin may contribute to weight loss, losing 30 pounds solely from metformin is uncommon. The effects on weight are mild, and healthier lifestyle changes such as improving diet and increasing physical activity are often more effective.
It’s also being considered for overweight patients with conditions like polycystic ovary syndrome (PCOS). After exploring Metformin’s potential for weight loss, you may have lingering questions regarding its effectiveness, safety, and suitability for your specific situation. While some products, like appetite suppressants, may effectively help you lose weight, they can also come with potential side effects. Other alternatives to Metformin include over-the-counter supplements and prescription medications that can assist in weight management. Always prioritize safety and effectiveness in your weight loss strategy and ensure you are well-informed before making any adjustments. Additionally, prioritizing sleep and managing stress can help you maintain a healthier weight while enhancing your overall well-being. Incorporating a combination of lifestyle changes, physical activity, and potential supplements may facilitate achieving your weight loss goals more effectively. These approaches can enhance your overall weight loss experience, providing diverse methods tailored to your needs. Many individuals looking to lose weight may benefit from exploring alternative strategies alongside medications like Metformin. It’s vital to have a thorough discussion with your medical provider to assess your unique health profile and determine the safest approach to weight management. In fact, those with heart problems or chronic respiratory issues should also exercise caution, as taking metformin could exacerbate these health concerns. Additionally, if you are pregnant or breastfeeding, consult your healthcare provider before considering this medication. This medication is generally not recommended for people with severe kidney disease, liver conditions, or a history of lactic acidosis, as it may worsen these conditions. Side effects and contraindications can be significant; hence, certain individuals should avoid metformin. Above all, some common side effects of metformin include gastrointestinal issues such as nausea, diarrhea, and abdominal discomfort.
Surprisingly, the mechanisms responsible for lowering body weight are unknown, even though weight loss alone improves glucose homeostasis in T2DM. Feeding metformin in pharmacologically relevant doses to mice on a high fat diet normalized HbA1c levels and ameliorated glucose tolerance, as expected, but also considerably slowed down weight gain. Clinically significant weight loss refers to losing at least 5% of your body weight, resulting in a reduced risk of heart disease. Liraglutide was initially approved by the FDA to help patients with T2D manage their blood sugar levels. This article explains how diabetic medications affect weight loss and which diabetic drug causes the most weight loss. This study aims to investigate metformin’s effects on aging-related diseases and overall lifespan, positioning it as a promising candidate for enhancing longevity and healthspan. Unexplained weight loss can occur in people who have Type 2 diabetes, but it’s more commonly found in people with Type 1. If you have Type 2 diabetes, your body doesn’t use insulin effectively and can’t transport the glucose to your cells. Measurements of height and weight were performed, and baseline blood samples were taken for fasting glucose, insulin, and HbA1c. Given the regulation of the cortisol-regenerating enzyme 11βhydroxysteroid dehydrogenase 1 (11βHSD1) by insulin and the limited efficacy of selective 11βHSD1 inhibitors to lower blood glucose when co-prescribed with metformin, we hypothesized that metformin reduces 11βHSD1 activity. No, insurance does not cover Semaglutide and Metformin for weight loss, even though obesity is linked to several health risks. So, to beat obesity, many people consider weight loss drugs like Metformin and Semaglutide. The right dosage and a combination of healthy eating and exercise can make metformin an effective part of your weight loss journey. Metformin can be a helpful tool for weight loss, especially for individuals with underlying conditions like Type 2 diabetes, PCOS, or insulin resistance. While metformin can support weight loss, it’s most effective when used in conjunction with a healthy lifestyle. If you’re using metformin specifically for weight loss, your doctor will likely start you on a lower dose and gradually increase it to avoid side effects.
Consider the story of John, a middle-aged man from Chicago struggling with type 2 diabetes and weight management. For many North American residents grappling with weight issues alongside diabetes, this question is more than academic; it’s a beacon of hope. Additionally, metformin decreases the amount of sugar your liver releases into your bloodstream and can reduce the absorption of sugar from your intestines. This is crucial because, if left unchecked, elevated blood sugar can lead to a host of complications, including heart disease, nerve damage, and even vision problems. Our findings may stimulate research into alternative weight management strategies for older women with ORCs that are effective and do not carry the same potential risks. However, cancer treatment modality is likely to modify the observed effects rather than simply confounding them. The available data did not provide sufficient information on these aspects, and as such, variability in patients’ exposure to these medications could not be assessed. While the focus of this study is on pre-diagnostic exposure, it does not account for the duration, dosage, or adherence to metformin or WLM. We managed immortal bias effectively by defining appropriate time windows for metformin and WLM exposure and limiting our analysis to patients diagnosed with ORC after the follow-up period began and who survived at least six months after ORC diagnosis. Our findings suggest a concerning association between pre-diagnostic use of metformin and WLM with increased mortality risk in older women with ORCs. A study analyzing data from 1999 to 2010 revealed a remarkably low prescription rate for WLM, with only 2% of obesity-related visits mentioning such medications66. Clinicians generally perceived metformin as well-tolerated and a safe option for managing diabetes. The complicated relationship between obesity, weight loss and management, and cancer mortality has been a subject of ongoing research. Insulin resistance and metabolic dysregulation may impair response to chemotherapy, while WLM use may alter drug metabolism, nutrient absorption, or immune function, potentially affecting treatment efficacy. Additionally, the discrepancy between our findings and prior studies reporting favorable survival outcomes with metformin use may reflect differences in model specification and covariate adjustment. Although IPTW using propensity scores was employed to balance observable baseline characteristics, this approach does not account for unmeasured confounding, including factors such as diabetes duration, glycemic control, body mass index (BMI), functional status, or frailty. Individuals prescribed metformin or WLM often have comorbid conditions that independently confer elevated mortality risk. The observed association between pre-diagnostic use of metformin or WLM and increased mortality risk may be driven by underlying clinical and methodological factors. Our findings align with a minority of studies reporting adverse cancer outcomes linked to metformin use14,15, they suggest a poorer cancer prognosis than is typically reported in the majority of the literature. The mechanisms by which metformin and WLM influence cancer mortality are likely complex, involving both direct and indirect effects.
Being positively charged, the drug accumulates in cells and, further, in the mitochondria because of the membrane potentials across the plasma membrane and the mitochondrial inner membrane . (1) Uptake of metformin into hepatocytes is catalysed by the organic cation transporter-1 (OCT1) . Note that the possible effect of metformin on mitochondrial glycerophosphate dehydrogenase has not been included. The most intensively studied mitochondrial action of metformin is the inhibition of Complex I of the respiratory chain 14, 16, which suppresses ATP production. Given that gluconeogenesis is an energy-intensive process (consuming six ATP equivalents per molecule of glucose synthesised), hepatocytes need to balance the demand for ATP with supply, with the latter primarily provided by mitochondria. The human pharmacokinetic data point to the liver, kidney and intestines as the key target organs of metformin and in this review we will primarily focus on the liver and intestines, particularly when referring to the beneficial impact of metformin on metabolism and inflammation. Metformin, metformin accumulation was observed in the pancreas and adipose tissue at a concentration of approximately half that seen in the liver ; how this translates to humans is unclear. Plasma concentrations of metformin in humans are typically in the low micromolar range (e.g. 8–24 μmol/l) but are 30–300 times higher in jejunal samples . Following oral dosing of immediate-release metformin in humans, approximately 70% of the dose is absorbed from the small intestine with the remainder passing into the colon before being excreted in faeces . Chemically, (a) galegine (also known as isoprenylguanidine), is an isoprenyl derivative of guanidine, while (b) metformin (dimethylbiguanide) and (c) phenformin (phenethylbiguanide) are biguanides containing two coupled molecules of guanidine with additional substitutions In this brief review, we summarise the current evidence highlighting how metformin’s benefits are likely to be caused by a variety of molecular mechanisms. It was established as a safe and effective therapy before detailed mechanistic studies became possible and, despite its clinical use for 60 years, its molecular mechanisms of action remain much debated. Chemically, galegine is an isoprenyl derivative of guanidine, while metformin and phenformin are biguanides containing two coupled molecules of guanidine with additional substitutions (Fig. 1). At about the same time, two synthetic derivatives of galegine, metformin and phenformin, were first synthesised and tested, although they were not introduced to clinical use until the 1950s . Galegine was tested as a glucose-lowering agent in humans in the 1920s but was found to be too toxic 1, 2. In the last 10 years, we have moved from a simple picture, that metformin improves glycaemia by acting on the liver via AMPK activation, to a much more complex picture reflecting its multiple modes of action.
Effectiveness of metformin on weight loss in non-diabetic individuals with obesity. Some ingredients like berberine have shown metformin-like effects in early studies, but they lack the same level of clinical research and regulatory oversight. Many people observe changes in appetite, energy, or weight within 8–12 weeks, especially when combined with healthy lifestyle habits. While it’s FDA-approved for managing type 2 diabetes, its use in supporting healthy weight, balanced energy, and reproductive health is gaining traction in wellness and preventive care settings. Some observational studies suggest that metformin may support heart health by promoting lipid balance and healthy vascular function. It also enhances the body’s cellular response to insulin, allowing for more effective glucose uptake into muscle and fat cells. Talk to your doctor today to see if it’s a fit for your health and weight loss journey. Read more under metformin dosage for weight loss in non diabetics above. ⭐ “I had no weight loss at first, but then pounds just started dropping.”— feedback from metformin weight loss Reddit discussion Compare that to newer drugs, and you’ll see why many people give metformin a try first.
The highest difference was shown in the study conducted by Kooy et al. with the difference of −2.91 mmol/L. The only study that showed an increment in the cholesterol level was the study conducted by Gillani et al. Similar to the findings of Hadigan et al., we did not observe any significant changes in lipid profile in different studies that this systematic review focused on and reviewed. This systematic review and meta-analysis showed discrepant results about the influence of metformin on lipid profile as a whole. The data were extracted from all the six studies to analyze the overall effect. Two of the studies did not have the same trial timing as all groups. Four studies were outcome assessor blinded, four of them were co-interventions avoided or similar, and three utilized intention to treat. Only one of the studies did not report allocation concealment. Overall nine studies of the 11 were used for qualitative synthesis, and six14,15,16,17,18,19 were used for meta-analysis and the rest three studies were excluded because the studies did not have the same trial timing for all groups. A total of 173 studies were screened, assessing for the eligibility of these titles and abstracts resulted in 11 publications that met our inclusion criteria for use in this systematic review and meta-analysis. The weighted mean difference and the 95% confidence interval (CI) were calculated for each outcome. A standardized data extraction form was used to evaluate the quality of identified studies. Studies with three or more bias risks were considered ineligible for data synthesis and analysis. In the second step of screening, duplicates were removed to avoid inappropriate studies and studies that were not in the aforementioned eligibility criteria. Exclusion criteria included patients who were pregnant, under screening of lactation, fertile females who were not using anticonception, patients with major cardiovascular illness, a history of heart failure, and diabetic complications, which include serious brain, kidney, lung, liver, or heart complications. A wide age range was included, as type 2 diabetes may develop at any age.
Research suggests that people who are obese may have fewer short-chain fatty acids in their gut — which affects many aspects of our health, including weight. Metformin may influence lipid metabolism by breaking down and using stored fat for energy, resulting in a reduction in body fat and, subsequently, weight loss. By regulating these hormones, metformin may assist in decreasing the appetite and promoting feelings of fullness, leading to reduced calorie intake and potential weight loss. Since then, studies have found that metformin promotes appetite regulation, resulting in a lower food intake and a decreased body mass index. There are several factors that could potentially help some people lose weight while taking metformin. Insulin is a hormone that occurs naturally in our bodies and is responsible for regulating blood glucose levels. Metformin is a widely prescribed medication that falls into a class of drugs called biguanides — an oral group of medications used to treat type 2 diabetes. "The fact that metformin and sprint exercise affect your body weight through the same pathway is both weird and interesting," Long said. "Now we know that it is acting through the same pathway as vigorous exercise to reduce hunger. Understanding how these pathways are controlled may lead to viable strategies to lower body mass and improve health in millions of people." \"The fact that metformin and sprint exercise affect your body weight through the same pathway is both weird and interesting,\" Long said. The researchers found that obese laboratory mice given metformin had increased levels of lac-phe in their blood.
If a patient has HIV/HBV co-infection, Dovato alone is generally not recommended as the primary treatment for Hepatitis B because resistance to Lamivudine can develop quickly. While current data suggests the risk is low, counseling is recommended for individuals of childbearing potential. Hepatic function should be monitored closely for several months in co-infected patients who discontinue Dovato. This happens because the Hepatitis B virus, which was suppressed by Lamivudine, can reactivate aggressively once the drug is stopped. Dovato carries a Boxed Warning regarding the risk of Hepatitis B exacerbation. The medication combines the potency of an Integrase Strand Transfer Inhibitor (INSTI) with the established efficacy of a Nucleoside Reverse Transcriptase Inhibitor (NRTI). Unlike traditional three-drug or four-drug regimens, Dovato is a complete, two-drug regimen (2DR) contained within a single daily pill. Dovato 50 mg/300 mg Film-Coated Tablets represent a significant shift in the paradigm of HIV treatment. You would get a baseline cardiac evaluation before starting which medication? High fever (103+), cardiovascular effects (tachycardia, heart failure, angina, MI), and CNS effects (agitation, restlessness, delirium, progressing to coma).
For those seeking enhanced weight loss effects, metformin 850 mg may be considered under medical supervision. Metformin, a medication primarily used to manage type 2 diabetes, has shown promise as a weight loss aid. For individuals looking to explore metformin's potential as a weight loss aid, this dosage may provide more significant effects. While it is primarily used to treat diabetes, studies have shown that this dosage may also lead to weight loss, particularly in overweight or obese individuals. Metformin 500 mg is a common starting dose prescribed to individuals with type 2 diabetes to help manage blood sugar levels. Metformin, a widely prescribed medication for type 2 diabetes management, has garnered attention for its potential benefits in aiding weight loss. Preclinical and clinical studies have demonstrated that treatment with antipsychotics and metformin had effects on the gut microbiota and the brain. In women with PCOS, metformin improved glucose homeostasis accompanied by decreased serum luteinizing hormone and thyrotropin levels, which indicated that metformin treatment may have an impact on pituitary activity (Billa et al., 2009; Krysiak and Okopien, 2015). Compared to vehicle-treated rats, body weight, blood glucose, and dopaminergic receptor expression in the cortico-mesolimbic system were higher in the olanzapine-treated Roman high-avoidance rats; in contrast, the Roman low-avoidance rats showed no differences in these measures. The other RCT was conducted in children and adolescent patients who had more than 10% increases in body weight after at least 1 year of SGA treatment. One of these studies demonstrated that metformin can prevent olanzapine-induced weight gain and insulin resistance in severe schizophrenia or schizoaffective adults (Baptista et al., 2006). However, patients who receive SGAs are at risk of metabolic dysfunctions, which may induce severe disease e.g., type 2 diabetes (T2D), cardiovascular disorders, and obesity, decrease patient compliance, and increase health costs (Lochmann van Bennekom et al., 2013; Werner and Covenas, 2014). While extrapolating this information to patients with T2DM may need further clinical studies, it is likely that lack of hypoglycemia in patients with T2DM treated with metformin is explained by enhanced hepatic glucose production due to increased glucagon secretion. This study thus offered insight into the effects of metformin in individuals with prediabetes.
It is found that women with PCOS have 30% – 40% more BPA in their blood compared to women without PCOS. She sought help from a PCOS weight loss specialist at Fitelo, who designed a personalized home-based diet plan, taking into account her preferences. So, consult your doctor before starting with Metformin for weight loss in PCOS. Its main mechanism of action is improving insulin sensitivity and reducing insulin resistance, which are often underlying factors contributing to weight gain in PCOS. It has gained significant attention as a potential solution for weight loss in women with PCOS. Inform your healthcare professional about all your medications, herbal products, or supplements, as they may interact with Metformin. You can check our blog on mustard seeds for weight loss to boost your metabolism. If you experience persistent or severe side effects, inform your healthcare professional. Stay in touch with your healthcare professional for regular check-ups to monitor your progress and any potential side effects. Be mindful of portion sizes to consume appropriate calories for weight loss. Some people may experience minimal or no side effects while taking Metformin for PCOS management. Metformin, a commonly prescribed medication for PCOS, is known to have some potential side effects.
It does have some side effects, but they tend to be mild. These are all situations where the risk of lactic acidosis increases. Metformin is not recommended for people with advanced kidney disease, significant liver impairment, or a history of lactic acidosis, Gidwani says. That combination of an opioid antagonist drug and an anti-depressant reduces cravings and hunger. That's because metformin's mechanisms of action are centered in the GI tract. You'll know if the metformin is an extended-release version because the brand name will have XR after it. The dosage you end up taking will depend on your particular health needs and what you're trying to address, says Lake. Metformin is a generic name for a drug that can either be prescribed on its own or under a brand name that contains other active ingredients. The combined effect of metformin and mirabegron on diet‐induced obesity. We also thank Prof. Lina Zhao (Sun Yat‐sen University, China) and Prof. Gang Shu (South China Agricultural University, China) for their assistance in body composition measurement. The body composition of mice was determined using an animal NMR system (MesoQMR23‐060H‐I, China) at the end of the experiment. The chosen dosages of Met and Mir were based on the previous literature.19, 27 All drugs were dissolved or suspended in the Veh, 0.4% (w/v) carboxymethylcellulose sodium solution (CMC‐Na, Macklin). Our findings provide a novel route to managing obesity where both EI and EE can be modulated using pre‐existing drugs. Furthermore, safety in human subjects will need to be carefully assessed in clinical trials before Met/Mir treatment can be offered to individuals with obesity. Considering that the majority of anti‐obesity pharmacotherapies failed to reach the market due to unwanted side effects as opposed to poor efficacy, it was crucial to evaluate the potential adverse effects of Met/Mir therapy. The exact mechanism by which Met/Mir improves glucose homeostasis remains an interesting new topic warranting future investigation. However, Met/Mir may also have other mechanisms to modulate insulin responsiveness that may explain this additive effect. Lipolysis in eWAT was further differentially regulated, exhibiting either an increase or no change in lipolysis from Met/Mir treatment.
Our review focuses on the rationale for their use, with advantages and disadvantages; on combinations often used, with or without studies; and on new perspectives of combinations being studied mainly by the pharmaceutical industry. Drugs combinations can be an option for its treatment but, although widely used in clinical practice, very few data are available in literature for its validation. If you’re still not seeing positive changes, review the instructions on your prescription—and then reach out to your healthcare provider. Vitamin B12 deficiency is another common side effect of metformin. Eating low glycemic foods and avoiding high-fat meals can help to minimize any GI side effects, she adds. “Side effects like nausea and acid reflux are common,” Dr. Granados says. “You can expect to see improvements in A1c after two to three months of using metformin,” Dr. Granados adds. That’s why it’s also important to monitor your A1c levels if you’re diabetic. “Weight loss with metformin is modest and slow,” Dr. Granados says. That can be one of the telltale signs metformin is working. But what are the signs metformin is working?
Metformin represents a valuable tool for weight management, particularly in individuals with type 2 diabetes or insulin resistance. Higher doses tend to produce more significant weight loss, but have a trade-off of potentially more side effects. After about nine months, once the majority of weight loss is achieved, the dose is typically reduced to 1,500mg per day to maintain weight loss and minimize side effects. Metformin dosing for weight loss typically starts with a low dose to minimize gastrointestinal side effects. Metformin, primarily known for its efficacy in managing type 2 diabetes, has gained attention for its potential benefits in weight loss and longevity. Hot, humid weather can affect your blood sugar levels and even cause dehydration or heat stroke Parents are often the first to notice unusual weight loss in a child with Type 1 diabetes. Type 1 diabetes has a similar pattern, but instead of being unable to use insulin, your body stops producing it altogether. Read on to find out why diabetes causes weight loss and what to do. Ultimately, this dramatic weight loss could be an early sign of diabetes. If this severe and unexpected weight loss is taking place, talk to your healthcare provider immediately. Metformin increased the Ra of D3-cortisol (Ra D3-cortisol, a specific measure of whole-body 11βHSD1 activity) compared with placebo (both groups) and gliclazide (ODM group only) (Figure 2B). Additional mechanisms contributing to the glucose-lowering effect of metformin have been proposed, such as the organic cation transporter Oct1, which enhances the action of metformin in the liver, whereas metformin may antagonize the effects of glucagon (reviewed in Ref. 3). Metformin is the mainstay of treatment in obese patients with type 2 diabetes mellitus (T2DM), yet the mechanism of action remains unclear. Obese men with and without type 2 diabetes received 28 days of metformin or placebo then underwent a deuterated cortisol infusion to measure 11βHSD1 activity. Metformin increased whole-body cortisol regeneration by 11βHSD1 in both groups compared with placebo and gliclazide and tended to increase hepatic 11βHSD1 activity. Also, taking Metformin for weight loss is regarded as an off-label use because it is approved for diabetes. While Metformin isn’t approved for weight loss, studies suggest 1500 mg may reduce weight. Patients who lost over 5% of body weight in the first year maintained an average 6.2% weight loss over 6 to 15 years.
ILS participants also had the lowest 2-hour plasma glucose levels over time (Fig. 2D). Model 1 included the baseline covariates sex, age at baseline, race/ethnicity, treatment, baseline weight, and baseline distance away from the SMA line. In participants with the same secretion compensation levels (same distances away from the baseline SMA line), the distances along the baseline SMA line allow the quantification of increase in insulin secretion per decrease in insulin sensitivity (9). Distances away from the baseline SMA line represent measures of adequacy of insulin secretion compensation in response to changes in insulin sensitivity and relate to the Stumvoll et al (8) measures of homeostatic response to glucose, but without assuming a hyperbolic relationship (9). Another purpose was to investigate factors affecting the movements of points over time with respect to the baseline SMA line, since the position of the points with respect to the curve representing beta-cell function determines the risk of developing diabetes (2, 9). We estimated longitudinal associations of weight change with trajectories of insulin secretion compensation and secretory demand estimated as previously described (9). These were compared by first checking if lines for the same treatment group but at different yearly visits shared a common slope with a likelihood ratio test (17); if so, we used a Wald test for common intercepts (17). Surrogate indices of insulin secretion and sensitivity were calculated from plasma glucose and serum insulin concentrations measured at fasting (G0, I0) and 30 minutes (G30, I30) during an OGTT. Longitudinal variables included weight change from baseline, G0, G30, G120, I0, I30, ISI0, CIR30, and measures of insulin secretory demand and secretion compensation described below. These indices are highly correlated with the gold standard measures of insulin secretion and sensitivity (15) and were used in the previous DPP analysis (11).Baseline covariates included age, sex, race/ethnicity, and DPP treatment assignment. We analyzed data from 2931 participants with baseline FPG Surrogate indices of insulin secretion and sensitivity were calculated from plasma glucose and serum insulin concentrations measured at fasting (G0, I0) and 30 minutes (G30, I30) during an OGTT. We conducted a secondary analysis using data from the DPP repository (14), which contained information on 3081 of the 3234 original participants randomized to the ILS, metformin, or placebo group. The primary outcome was diabetes development which was diagnosed using an annual oral glucose tolerance test (OGTT) or a semiannual FPG test using the American Diabetes Association 1997 criteria (2-hour plasma glucose ≥11.1 mmol/L or FPG ≥7.0 mmol/L), with confirmation by a repeat test. Finally, we studied the time-dependent associations of insulin secretion compensation and secretory demand with the transition from IGR to type 2 diabetes development.

As described by Baker et al. in their systematic review and meta-analysis, dapagliflozin also decreases systolic blood pressure by 4–5 mmHg and results in a 1.7 kg weight loss (95% CI 1.33 to 2.11) . Dapagliflozin is a selective sodium-glucose cotransporter type 2 inhibitor (iSGLT2) that blocks glucose resorption in the proximal tubule of the kidney, thereby increasing urinary glucose excretion and reducing blood glucose levels. This randomized phase IV clinical trial will include patients with diabetes or prediabetes who are between the ages of 18 and 60 years and exhibit grade III obesity (defined as body mass index ≥ 40 kg/m2). Metformin and dapagliflozin are two drugs approved for the treatment of diabetes. The treatment goals and duration of therapy should be clearly defined, in particular, in overweight-obese women with PCOS and normal initial glucose homeostasis where its long-term use is currently more difficult to advocate. The next important question is for how long metformin should be applied to reach the homeostasis that can sustain weight and glucose metabolism after metformin withdrawal. We encourage future designs to investigate the stabilization of BM through the years as one of the main treatment benefits of long-term treatment with metformin in overweight-obese PCOS. This offers very important and rarely available insight into the long-term longitudinal follow-up in this subset of patients that have not been, in general, characterized as candidates for metformin treatment until the latest recommendations update (7). The main strength of this study is the long-term longitudinal follow-up assessing the effectiveness of treatment with metformin in real life setting that is insufficiently studied in PCOS. One of the longest retrospective study with 50 patients followed by a mean treatment period of 43.3 months demonstrated a 11-fold decrease in the annual conversion rate from NGT to IGT and complete prevention of the development of T2D (40). Nonetheless, considering that these women are at high risk for developing T2D (39, 40), it has been suggested that they will benefit from metformin therapy in case of glucose intolerance (7). Prospective studies investigating the impact of metformin on T2D risk specifically in women with PCOS are lacking (6). In an elegant study by Legro et al., there was a nearly two-fold increase in the rates of conversion for subjects with PCOS and baseline NGT compared with the reference population, but there was also a significant chance for a spontaneous reversion rate to normal glucose tolerance (38).
Anyone having big reliability issues with Freestyle Libre 2 Plus sensors? Type 1 Diabetes
- The active pharmaceutical ingredient in this medication is Dapagliflozin (typically present as dapagliflozin propanediol monohydrate).
- Multiple mechanisms underlie the weight loss-inducing and health-promoting effects of metformin.
- The mechanism of action for the antiepileptic activity of this medication is not totally clear, but it is believed that it is linked of sodium and calcium channels blockage.
- Metformin does not cause dramatic weight loss, but it can cause small, steady changes.
- Obesity induces various degrees of inflammation in adipose tissues, pancreatic islets and the liver, which contributes to hepatic steatosis, systemic insulin resistance and progression towards T2DM, non-alcoholic steatohepatitis (NASH) and cardiovascular diseases152–154.
- As part of your subscription and as medically indicated, physicians prescribe medications, and recommend supplements that are delivered to you from the comfort of your home.
- Metformin reduces the amount of glucose produced by the liver and improves insulin sensitivity.
- After the first year, about 29% of people on metformin lost at least 5% of their weight, while about 63% of those in the lifestyle group reached that goal.
These effects include modulation of different points of cancer timeline, weight reduction, cardiovascular health, thyroid diseases, polycystic ovaries disease and many other medical conditions. These side effects often improve over time as the body adjusts to the medication. In addition to its benefits for blood sugar control and weight management, Metformin can help regulate menstrual cycles and improve ovulation. The weight loss effects are typically modest and vary from person to person. It is important to note that while Metformin can aid in weight loss, it is not a miracle weight loss drug. Several studies have shown that Metformin can lead to modest weight loss in some individuals. The key to success lies in a personalized approach, considering an individual’s unique health profile, lifestyle, and weight loss goals. This synergy promotes a healthier lifestyle and aligns with the long-term management of diabetes, showcasing metformin’s dual benefits. What sets metformin apart is its reinforcement of the importance of a comprehensive approach to weight loss. Are you pondering whether metformin could be the missing piece in your weight loss puzzle? However, embracing a personalized approach to health and weight loss is crucial. Women with PCOS, a condition often accompanied by insulin resistance and weight gain, may also find metformin beneficial. This weight loss gradually emphasized metformin’s role in facilitating a sustainable weight reduction rather than offering a quick fix. One of the most significant ways metformin may contribute to weight loss is its influence on appetite. Metformin’s journey from a diabetes management tool to a potential ally in weight loss is paved with intriguing scientific discoveries. “I started metformin for my diabetes,” she shares, “but over time, I noticed my cravings reduced, and I was losing weight without the intense struggle of before. Participants not only saw improvements in their blood sugar control but also experienced a gradual weight reduction. Consider the findings of a landmark study that followed individuals with type 2 diabetes on metformin. Metformin’s secret to promoting weight loss lies in its multifaceted approach to the body’s metabolism and appetite regulation. Understanding metformin’s role in weight loss begins with a review of the scientific evidence.
This means that your cells can better take in and use sugar from your blood, lowering blood sugar levels. Unlike medications that increase insulin production in the pancreas, metformin takes a different route. Metformin’s mission in the medical field is primarily to combat high blood sugar levels. Compare metformin’s subtle powers against dedicated weight loss pharmacological contenders. This common diabetes medication uniquely improves insulin sensitivity, which regulates appetite and fat storage. Touted as magic, metformin’s multiplying benefits now expand to gradual, sustainable weight loss for some. Metformin and weight loss medication impact on survival outcomes in older women with obesity-related cancers. However, given the substantial burden of ORC and increasing interest in weight loss treatments, further investigation into the safety and prognostic implications of WLM is warranted. In summary, this population-based cohort study employed multivariable Cox regression and competing-risks models with IPTW-PS to investigate the association between pre-diagnostic use of metformin and WLM with mortality outcomes in older women with ORC, including BrCa, CRC, ECa, and OCa. These studies should investigate how metformin and WLM influence tumor biology, including their impact on metabolic alterations, inflammation, and insulin resistance. Firstly, large-scale epidemiological studies should examine the long-term effects of pre-diagnostic metformin and WLM use on overall survival and cancer-specific outcomes, considering time- and dose-response.
Stopping metformin abruptly can lead to a rapid regain of weight, as the body’s insulin sensitivity and glucose metabolism return to pre-treatment levels. In the current study, we aimed to investigate the long-term effects of metformin on blood glucose control in non-obese Japanese patients with type 2 diabetes mellitus. A medication primarily used in the management of type 2 diabetes, metformin helps improve insulin sensitivity and lower blood sugar levels. While metformin is not classified as a weight loss drug, it can help people who need to manage both weight and blood sugar. In addition, in a placebo-controlled trial in patients with prediabetes, treatment with metformin reduced the concentrations of the neutrophil-derived extracellular trap (NET) components elastase, proteinase 3, histones and double-strand DNA, independently of its effect on normalizing glucose levels160. Similarly, randomized controlled trials in both obese non-diabetic adults and non-insulin dependent women with diabetes have reported that, in addition to reducing body weight, metformin treatment leads to a greater reduction in energy intake than placebo treatment. For example, we found that, compared to placebo-treatment, metformin reduced body mass index (BMI) by an average of 1.09 kg/m2, and body weight by 3.38 kg, in 6–12y overweight and insulin-resistant children over a 6-month period . While the exact mechanisms behind Metformin’s potential weight loss effects are not fully understood, its ability to regulate blood sugar levels and improve insulin function may play a role. Metformin, a medication primarily prescribed to help individuals manage their blood sugar levels in cases of type 2 diabetes, has garnered attention for its potential side effect of weight loss. It was repeatedly demonstrated throughout the PIONEER clinical trials (table 3) that the decrease in both HA1c and body weight was significantly greater with oral semaglutide when compared with a placebo.31–35 The superiority of oral semaglutide was also observed when compared with other oral antidiabetic medications, such as sitagliptin30 36 and empagliflozin.26 This superiority was also found in studies comparing oral semaglutide with other agents from the same drug class (dulaglutide37 and liraglutide33 34). This medication treats high blood sugar levels by increasing insulin sensitivity and reducing glucose production in the body. Although the weight loss produced is small, metformin treatment may hold promise as a method to prevent or delay the appearance of impaired glucose homeostasis in children at high risk for the development of type 2 diabetes.
Metformin and Lifestyle Change
These side effects are usually mild and temporary, but in some cases, they can be more severe. Obtaining Metformin online requires a prescription from a licensed healthcare provider. Always follow the prescribing information and the advice of your healthcare provider regarding how to take Metformin. Therefore, it's important for patients to have their kidney function tested before starting Metformin and periodically thereafter. This risk is higher in individuals with kidney disease, severe dehydration, or liver problems. These side effects often diminish over time and can be minimized by taking Metformin with food. The most common side effects include gastrointestinal issues such as nausea, vomiting, diarrhea, and stomach upset. The U.S. Food and Drug Administration (FDA) approved Metformin in 1994, underscoring its safety and efficacy for managing diabetes. In animal models, Metformin has been observed to increase the effectiveness of leptin in controlling appetite and reducing fat, suggesting potential mechanisms through which it may aid in weight management in humans. Metformin comes in both immediate-release and extended-release formulations, allowing for flexibility in dosing and minimizing side effects. Taking Metformin when not diagnosed with diabetes should be approached cautiously and is generally not recommended without medical advice. Yes, incorporating lifestyle changes is essential when using Metformin for weight management. This risk is particularly relevant for individuals with kidney impairment, liver disease, or severe dehydration. In rare cases, Metformin can lead to a serious condition called lactic acidosis, characterized by an accumulation of lactic acid in the bloodstream.
To achieve significant weight loss, it is essential to combine Metformin with a healthy diet and regular exercise. These effects may result in a reduction in calorie intake and an increase in calorie burning, leading to weight loss. Metformin is an antidiabetic medication that has gained attention in recent years for its potential to aid weight loss. Regular exercise can improve insulin sensitivity and help the body use glucose more efficiently. Therefore, Metformin may be a useful medication for people with type 2 diabetes or other metabolic conditions. Combining Metformin with a healthy diet and regular exercise routine can improve weight loss outcomes. The study’s results indicated that participants lost an average of 5.5% of their initial body weight over the two-month period. However, some participants did not experience significant weight loss or experienced side effects such as gastrointestinal discomfort. The study involved a group of 20 participants, all of whom were overweight or obese and had a body mass index (BMI) of 25 or higher. While there are several ways to achieve weight loss, medications such as Metformin have gained attention for their potential to aid weight loss. You will need to monitor your blood sugar carefully so your doctor will be able to tell how well metformin is working. Your doctor will order certain tests before and during treatment to check how well your kidneys are working and your body's response to metformin. Drinking alcohol increases your risk of developing lactic acidosis or may cause a decrease in blood sugar. Taking certain other medications with metformin may increase the risk of lactic acidosis. However, this does not mean that all people taking the antidiabetic medication will experience any or all of the following side effects. Like almost all medications, metformin will affect different people in different ways. Additionally, the use of JMs for time-to-event and multivariate longitudinal data allowed us to relate the underlying true values of insulin secretion compensation and insulin secretory demand with the risk of diabetes at any point in time without having to assume that these variables only changed at the time of yearly visits. This study investigated the progression of insulin secretion deficiency and decreased insulin sensitivity during the transition from IGR to diabetes and allowed us to understand how these factors interact with one another and what affects their interaction. By contrast, at the first examination with diabetes, insulin sensitivity and secretion had moved to the left and down, in other words, worsened, from their baseline relationship in all treatment groups (Fig. 4B). 1A, 1I, and 1J indicated that these 2 treatments decreased weight from baseline and insulin secretory demand and increased compensatory insulin secretion.
Obese Individuals without Diabetes
Circulating metformin concentrations are typically 10–40 μm in humans (23), whereas hepatic concentrations can reach 100–200 μm in rodents (24), meaning our in vitro metformin concentrations encompassed the physiologically relevant range. Although the ODM group was older, which could be a potential confounder, we have not observed any increase in cortisol regeneration by 11βHSD1 with age in previous studies (6, 16). In both the hepatocytes and adipocytes, the highest metformin concentration (10 mm) decreased cortisol generation by 11βHSD1. Placebo-phase data in OND and ODM groups were compared using the unpaired t test. The Ra of cortisol (Figure 2A) and rate of D4-cortisol clearance (Table 1) were unaltered by either treatment and were not different between ODM and OND groups. Placebo-phase data for OND and ODM groups were compared using unpaired t tests. Phases were compared using paired t tests in the OND group and repeated measures ANOVA with post hoc Fisher's LSD testing in the ODM group. One of the OND subjects developed transient diarrhea during the metformin phase. Subjects in the ODM group were older and had higher fasting glucose and HbA1c than the OND participants (Table 1). The volume of distribution for cortisol was taken as being 12 L, as in previous studies (12, 18). Transitions monitored for were m/z 130.1 → 60.1 and m/z 136.2 → 60.1 for metformin and D6-metformin, respectively, and m/z 324.2 → 153.1 and m/z 497.1 → 372.1, for gliclazide and D3-glyburide, respectively. Gradient elution was from 20–90% in B, where metformin and D6-metformin eluted at 1.1 minutes and gliclazide and D3-glyburide eluted at 2.0 and 2.3 minutes, with a total run time of 5 minutes. Metformin and gliclazide were extracted from plasma (100 μL) using an SLE+ plate (Biotage) after enrichment with D6-metformin and D3-glyburide as internal standards (200 ng). Three days after plating (hepatocytes) or after completion of differentiation on day 12 (adipocytes), cells were cultured for 24 hours in vehicle or 100 nm, 1 μm, 10 μm, 100 μm, 1 mm, or 10 mm metformin hydrochloride (Sigma). To measure whole-body 11βHSD1 activity, cortisol (containing 20% 9,11,12,12-2H4-cortisol D4-cortisol; Cambridge Isotopes) was infused at 1.74 mg/h for 4 hours after an initial 3.5-mg bolus (16). Transgenic mice overexpressing 11βHSD1 in adipose tissue or liver develop features of the metabolic syndrome such as obesity, glucose intolerance, and dyslipidemia (9, 10).
Victoza is a prescription medication that is FDA-approved to treat type 2 diabetes. Sesame offers an affordable and comprehensive online weight loss program to help you get started on your weight loss journey. Metformin is a prescription drug, meaning that you must have a written order from a licensed healthcare provider to get it. Talk to your healthcare provider about Wegovy for a more targeted treatment approach. There are several drugs used for the treatment of obesity on the market. Some atypical antipsychotic medications can cause weight gain. Metformin is also sometimes prescribed to treat antipsychotic drug-induced weight gain. This information will help reduce the risk of any interactions or adverse metformin reactions. Gastrointestinal side effects of metformin include diarrhea, bloating, stomach pain, gas, indigestion, constipation, and heartburn. While metformin is considered safe, it may cause side effects in some individuals.
However, the average weight loss in the liraglutide group (3.6 kg) was considerably greater than in the lixisenatide group (1.9 kg) . With lixisenatide and sitagliptin, the average weight loss at 24 weeks was 2.5 kg and 1.2 kg, respectively . GetGoal-O, a global, randomized, placebo-controlled, double-blind, phase 3 trial in which 350 patients were allocated at random to receive lixisenatide or a placebo, evaluated the efficacy and safety of lixisenatide in T2DM patients aged 70 years or older . In GetGoal-Duo 2, the effectiveness of lixisenatide was compared to prandial insulin administered once or three times each day . The 2-step titration approach was used to randomly assign 446 T2DM patients to either placebo or 20 μg of lixisenatide once daily . However, the average weight reduction was 0.2 kg in lixisenatide group and 0.2 kg gain in the placebo group without any significant differences . Moreover, GetGoal-P clarified the effectiveness and safety of lixisenatide in T2DM patients using metformin and pioglitazone . In this 24-week, parallel-group, double-blinded experiment, 855 T2DM patients were randomly assigned to receive either placebo or 20 µg of lixisenatide subcutaneously once daily (following the 2-step titration method) . GetGoal-S clarified the effectiveness and safety of lixisenatide in T2DM patients on metformin and a sulfonylurea . In GetGoal-X, a 24-week, randomized, open-label research, 634 metformin-using T2DM patients were randomly assigned to receive either 20 μg of lixisenatide once daily or 10 μg of exenatide twice daily .
In the prevention model, Met and/or Mir did not affect heart rate, systolic blood pressure (SBP), diastolic blood pressure (DBP), or mean blood pressure (MBP) (Figure S5A–D). Overall, Met/Mir treatment has an additive effect on BAT thermogenesis and scWAT browning but not on lipolysis, lipid oxidation, and browning in eWAT in DIO mice. Of note, unlike Met, Mir also increased UCP1 protein levels (Figure 8J), demonstrating an improved thermogenic capacity in eWAT. Mir‐treated eWAT displayed upregulated mRNA expression levels in panels of markers in thermogenesis, lipolysis, and fatty acid oxidation (Figure 8I–L). These data suggest that, unlike the limited changes in scWAT, eWAT is more responsive to Met treatment. The eWAT weight was reduced the most out of all the WAT depots, as shown in Figure 5K. Strikingly, similar to that in BAT, Met/Mir additively increased UCP1 proteins in scWAT (Figure 8F), which may contribute to enhanced scWAT browning and elevated whole‐body EE. Met/Mir led to a similar expression of key genes in WAT browning, lipolysis, and fatty acid oxidation when compared to Mir (Figure 8E–H). (M) A summary of thermogenesis, lipolysis, and fatty acid oxidation in BAT, scWAT, and eWAT after Met and/or Mir treatment in the treatment model. Metformin (Met)/mirabegron (Mir) has additive effects on brown adipose tissue (BAT) thermogenesis, fatty acid oxidation, and subcutaneous white adipose tissue (scWAT) browning in diet‐induced obesity (DIO) mice. To dissect how Met/Mir additively promoted weight and fat loss in DIO mice, we evaluated the expression of some key genes involved in thermogenesis, lipolysis, and fatty acid oxidation in BAT and WAT. Metformin (Met)/mirabegron (Mir) improves glucose metabolism in diet‐induced obesity (DIO) mice. Metformin (Met)/mirabegron (Mir) exerts an additive effect on energy expenditure (EE) in diet‐induced obesity (DIO) mice in the treatment model. Neither RER nor physical activity was altered by any treatment (Figure 6E,F). Strikingly, Met/Mir boosted EE to the highest of the four treatment groups, significantly higher than that induced by Met or Mir alone (Figure 6D). As expected, Met increased O2 consumption and CO2 release in DIO mice post‐drug administration (Figure 6B,C). Food consumption was reduced in DIO‐Met mice but was unchanged in DIO‐Mir and DIO‐Met/Mir mice when compared to their Veh counterparts (Figure 6A). Notably, BAT weight was significantly reduced in DIO‐Mir mice but not in DIO‐Met or Met/Mir‐DIO mice (Figure 5M). Less pronounced effects were seen in rWAT, with a marked reduction seen in the Mir and Met/Mir groups without a statistical difference between them (Figure 5L). Moreover, Met, Mir, and Met/Mir significantly reduced eWAT weight by 0.46 g (–21%), 1.00 g (–46%), and 1.39 g (–64%), respectively.
- This happens because your blood sugar is becoming more stable daily.
- The lifestyle intervention was thorough and structured and this kind of program is often not available to us for our patients.
- Moreover, in these studies it was shown that the 2-year and 5-year survival of patients using metformin is longer compared to non-users of metformin.
- These mechanisms facilitate better blood sugar control, making it a potential candidate for managing type 2 diabetes.
- One of the most significant ways metformin may contribute to weight loss is its influence on appetite.
- In non-diabetic individuals, metformin was shown to exert weak but beneficial effects on weight loss.
- Since high blood glucose causes the most severe complications in T2DM, researchers interested in understanding its mode of action mostly concentrated on the suppression of glucose production in the liver, which certainly occurs in vivo in patients upon treatment .
- The efficacy of pramlintide was evaluated in obese, nondiabetic subjects in a double-blind, placebo-controlled dose-ranging study .
- Standard or instant release (IR) tablets typically release metformin immediately upon digestion and usually lasts about 5 hours, so it must be taken 1-3 times a day, depending on your prescribed dosage.
The absence of a warning for a given drug or drug combination in no way should be construed to indicate that the drug or drug combination is safe, effective or appropriate for any given patient. Drugslib.com information has been compiled for use by healthcare practitioners and consumers in the United States and therefore Drugslib.com does not warrant that uses outside of the United States are appropriate, unless specifically indicated otherwise. Metformin passes into breast milk in non-significant amounts and has not been linked with side effects in any breastfed babies. The most common side effects are feeling and being sick, diarrhoea, stomach ache and loss of appetite. If you miss a dose of metformin, skip the missed dose and take the next dose at the usual time. Slow-release tablets work gradually and are usually prescribed if you are suffering from any side effects when taking the standard-release tablets. Please discuss your medications with your team prior to any tests or surgery. You may need to stop taking metformin before having surgery and certain medical tests. It is also safe to take during pregnancy, either alone or combined with insulin. If you have Gestational diabetes, Metformin is usually discontinued once you have delivered your baby. This can happen in pregnancy for some women with risk factors (NICE guideline Section 1.2.2) Metformin is a medicine used to treat Type 2 diabetes and Gestational (pregnancy) diabetes. When we administered the daily dose of metformin by gavage, as done by others, we also found increased ACC phosphorylation (Figure 5B), emphasizing that hepatic metformin action is highly dose dependent, as discussed recently . Breakdown of glucose-1-13C will yield one molecule of lactate-3-13C, which can be converted in the aldolase reaction into doubly-labeled glucose-1,6-13C, when by chance two labeled lactate-3-13C molecules are used for gluconeogenesis (Figure 6). We reasoned that metformin is an inhibitor of complex I of the respiratory chain, at least at high concentrations , , which are certainly reached in the intestine after oral administration . We first excluded the most trivial reasons like decreased food intake due to metformin's bitter taste, increased locomotion due to enhanced foraging activity, or malabsorption due to inhibition of active transport processes in the intestine. Indeed, liver tissue showed significantly lower levels of lactate at this stage (Figure 2C). In order to demonstrate this pathway directly, mice were given labeled glucose-1-13C in HFD. In order to substantiate this, metformin concentrations were analyzed and found to be 13 ± 5, 40 ± 13 and 37 ± 27 μmol/L after feeding HFD with 0.5%, 1% and 2.5% metformin, respectively. This demonstrates that at our feeding conditions, metformin obviously did not reach the liver in concentrations high enough to activate ACC phosphorylation and other downstream pathways.
On the other hand, chronic Metformin use was demonstrated to improve insulin ability to lower hepatic glucose production, positively affecting glycemic control. This effect is also achieved through facilitation of glucose uptake by peripheral tissues, especially muscle cells, via increased translocation of glucose transporter proteins 4, which increases insulin sensitivity. The primary mechanism of action of Metformin revolves around inhibition of gluconeogenesis by activating AMPK in the liver, eventually lowering blood glucose level. As recent observations and studies highlighted new evidence pertaining to diversity of its mechanisms, this article holistically presented the narrative linking Metformin to gastrointestinal effects and intolerance, referencing available evidence. Additionally, the use of Metformin in the treatment of inflammatory bowel disease demonstrated positive effects through its action on inflammatory pathways, intestinal barrier integrity and gut microbiota. LKB1 is required by Metformin to carry its action in the liver by facilitating the activation of the adenosine monophosphate (AMP) activated protein kinase (AMPK), which is responsible for glucose uptake by muscle tissue leading to lowering of blood glucose level.22,23 This pharmacological pathway that utilizes AMPK was found to affect lipid metabolism as well. Metformin, the widely recognized Biguinide, is a Guinide derivative derived from Galegine, which was historically extracted from a plant named Gallega officinalis.1–3 It is used for treatment of type 2 diabetes mellitus (T2DM), whose incidence is increasing yearly. If you have any questions about metformin not covered in this article or, you are looking for advice or treatment options for a loved one, get in touch today. But metformin doesn’t replace a healthy diet. However, metformin may also increase levels of the hormone leptin, so you feel full more quickly. Metformin also lowers insulin levels helping to reduce hunger. However, all medications have side effects. This formulation can allow a simpler dosing regime and reduce gastrointestinal side-effects, so many patients find it more acceptable. Metformin is also prescribed as a modified-release medication, which is gradually released into the bloodstream. It is commonly prescribed as a treatment for type 2 diabetes and gestational diabetes.
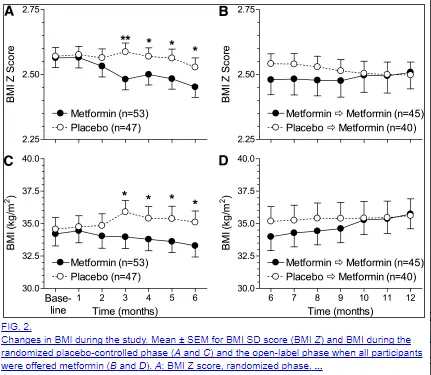
Discover if diabetes can be reversed and which steps to take to help manage your diabetes diagnosis. Learn about some common causes and symptoms of diabetic ketoacidosis (DKA), as well as effective prevention and treatment options. Discover why some researchers are exploring classifying Alzheimer’s disease as type 3 diabetes. Enrolling in the program means you’ll be working closely with licensed clinicians who will design a personalized plan to help you lose weight. With LifeMD, you can consult a healthcare professional via telehealth from the comfort of your home. Metformin carries a black box warning from the FDA which is the most serious warning a medication can get. Lactic acidosis may occur because metformin interferes with how lactate is metabolized in the liver. This symptom relates to the buildup of lactic acid in the bloodstream, which can be fatal. It’s unclear why this happens, but researchers believe it is because metformin interferes with how this vitamin is absorbed in the gastrointestinal tract. The drug is believed to affect the taste buds and alter the metabolism, causing a metallic taste. Some clinical trials have found that those taking metformin may experience a metallic taste in their mouth. However, it is believed that this is caused by how metformin impacts the appetite-regulating hormones in the brain.
Stomach-related side effects like diarrhea, nausea, and vomiting are fairly common, affecting up to about 30% of people who use metformin. After the first year, about 29% of people on metformin lost at least 5% of their weight, while about 63% of those in the lifestyle group reached that goal. It’s also the only medication recommended by the ADA for people with prediabetes to help delay or prevent the onset of type 2 diabetes. The study went further, showing that even a single dose of metformin can increase Lac-Phe levels. In two separate clinical trials, scientists found that metformin raises the levels of a hormone called GDF15 (growth/differentiation factor 15) in the blood. When insulin levels become more balanced, some people notice reduced hunger and less fat buildup. It’s not as dramatic as what’s seen with newer weight-loss drugs, but its effects are gradual, sustainable, and tend to last as long as you keep taking it. No, metformin is a prescription medication commonly used to manage type 2 diabetes. The research included all clinical trials investigating the effect of sitagliptin in obese or overweight adult patients with type 2 diabetes without any language restriction. Ideally, future studies will be designed to ensure clinical differences in weight loss as well as balanced use of other cardioprotective interventions. At present, most surgery is reserved for patients with more severe obesity, but these findings do provide further evidence of the benefits of weight loss in T2DM, and guidelines for the use of bariatric surgery may be updated in the near future 86. Therefore, while all available weight-loss drugs show some promise in the management of obese patients with T2DM, their exact role is as yet uncertain. These drugs have been primarily tested in patients with obesity alone, and to date, only one study has reported the use of lorcaserin in T2DM. The mechanism of weight reduction, by increasing urinary excretion of glucose, has created the perception that the weight reduction may be via water loss as well as lost calories, but studies have shown reductions in both visceral and subcutaneous adipose tissue 74,75. Among women who participated in the study, weight loss reduced the incidence of urinary incontinence at 1 year, although it did not improve resolution rates 62.
First, lactate and pyruvate concentrations increased, reflecting the enhancement of anaerobic glycolysis by metformin treatment . There was slight but significant decrease in RQ during post-prandial state by metformin treatment (Fig 4). RQ during light phase decreased significantly in metformin-treated rats compared to control rats. However, visceral fat volume was significantly decreased by the treatment of metformin while subcutaneous fat did not differ significantly between the groups (Table 1). Open circle and dotted line; before metformin treatment, closed circle and solid line; after a 2-week treatment by metformin. A, B; glucose, C, D; insulin, E, F; triglyceride, G, H; lactate. Glucose levels were determined in blood extracted from the tail before (0 min) and 15, 30, 60, 90 and120 min after an ip pyruvate injection. Blood glucose concentrations were measured with a handheld glucose meter (One Touch Ultra; LifeScan, Milpitas, CA) immediately after blood sampling. Plasma glucose, serum triglyceride levels were assessed according to the standard procedures. Metformin hydrochloride (1500 mg/day) was administered either 23 healthy subjects or 18 type 2 diabetic patients for 2 weeks. Thus, it is hypothesized that the administration of metformin promotes the reduction of body fat amount via the acceleration of fat oxidation and adaptive thermogenesis in vivo. Furthermore, it has been reported that metformin enhanced expressions of uncoupling proteins (UCPs), which are key enzymes of adaptive thermogenesis, both in animal study and in vitro culture study . Asian people, including Japanese, are therefore thought be much more likely to be affected by diabetes than people in western countries in the era of satiety. However, it is of interest that the prevalence of diabetes in Asian people is almost the same as that in Caucasian despite of their much lower BMI. Metformin treatment brought about a significant reduction of visceral fat mass compared to controls accompanied by an up-regulation of fat oxidation-related enzyme in the liver, UCP-1 in the brown adipose tissue and UCP-3 in the skeletal muscle. RQ during a fasting state was significantly decreased in metformin-treated rats compared to controls with no effect on EE. By the administration of metformin to SD rats for 2 weeks, plasma levels of lactate and pyruvate were significantly increased in both fasting and post-prandial states. Although energy expenditure (EE) did not change, baseline respiratory quotient (RQ) was significantly decreased and post-prandial RQ was significantly increased vice versa following the metformin treatment in both groups. Metabolic parameters and energy metabolism were measured during a meal tolerance test in the morning before and after the treatment of metformin.
- The treatment using additional agents was significantly more frequent in the individuals with diet therapy alone at baseline than in those using other glucose-lowering drugs in both non-obese and obese patients.
- Together, they help reduce the risk of long-term complications such as nerve damage, kidney issues, heart disease, and poor circulation.
- So, what are the signs that metformin is working for weight loss?
- Table 2 presents the results of the cross-sectional linear regression models for distances away from the baseline SMA line and distances along the baseline SMA line fitted at years 1, 2, and 3 to determine whether weight change explained the effects of ILS and metformin on both distances.
- When oxidative phosphorylation-dependent ATP production was inhibited by oligomycin, metformin still significantly increased the ATP production, suggesting a glycolysis-dependent mechanism.
- Combining Metformin with lifestyle changes can lead to better results, improved blood sugar control, and overall better health.
- Metformin treatment increased the expression of cellular proliferation and differentiation markers in brown adipocytes, alongside increased BAT mass224.
- Metformin can be a valuable addition to a weight loss regimen, particularly for individuals with type 2 diabetes.
Maximizing metformin's potential in fertility treatment requires a comprehensive approach that includes optimizing dosage and duration, lifestyle modifications, and managing common side effects. Foods that are rich in fiber, such as fruits, vegetables, and whole grains, can also help regulate blood glucose levels. A healthy lifestyle can improve insulin sensitivity, reduce androgen levels, and promote ovulation. Lifestyle modifications, such as diet and exercise, can significantly enhance metformin's effects on insulin resistance and fertility. In this article, we will explore how to maximize metformin's potential in fertility treatment, including optimizing dosage and duration, lifestyle modifications, and managing common side effects. So while turmeric may support your overall health, the evidence doesn’t show that it directly leads to major or rapid weight loss—and it isn’t a standalone solution. “Turmeric may support overall health, particularly through its anti-inflammatory properties,” adds Dr. Mann, “but the evidence doesn’t show that it directly causes weight loss. A meta-analysis of 14 studies published in Nutrition & Diabetes found that people taking curcumin lost about 4 pounds more than those taking a placebo. And, while studies suggest some promising ways that curcumin could help with weight loss, there isn’t enough research to prove its use over other evidence-based methods. Why do people think turmeric can help with weight loss? This is good news if you have type 2 diabetes, because high amounts of insulin can make it harder for your body to burn fat stores as an energy source. Metformin has become a very popular weight loss medication, and with good reason. Metformin is not approved by the FDA for weight loss purposes, but many doctors prescribe it anyway because they see how effective it can be at helping their patients shed pounds. Although metformin is generally considered safe, there are some side effects you should be aware of before taking this medication.
What is the main problem in Type 2 diabetes? There is absolute insulin deficiency. Decreases glucose production and increases storage as glycogen. What does insulin do in the liver? Treating anxiety and depression in individuals with IBS often requires a multidisciplinary approach, involving healthcare providers, therapists, and nutritionists. A healthcare provider or registered dietitian can help individuals develop a personalized diet plan that takes into account their unique needs and symptoms. As such, weight changes can vary greatly from person to person with IBS. In some cases, individuals may turn to comfort foods or overeat in an attempt to cope with symptoms, leading to weight gain. However, it’s important to note that weight gain can also be a symptom of IBS, particularly for those who experience constipation-predominant IBS. Keeping a food diary or symptom journal can help individuals identify triggers and patterns, which can inform treatment decisions. Some people may experience urgent needs to have a bowel movement, while others may struggle with straining during bowel movements. This can include dietary changes, stress management, and medication. The symptoms can be debilitating, affecting not only the digestive system but also overall health and wellbeing. Irritable Bowel Syndrome (IBS) is a chronic gastrointestinal disorder that affects millions of people worldwide. Get personalized dieticians and health coaches to help you live a healthly and fit life. As always, consult with a nutritionist if you have any questions or concerns about starting a new diet or management of your weight! Our team of healthcare professionals is here to help you reach your health and wellness goals! Moreover, you should always follow the directions on the label when taking metformin. It is important to talk to your doctor before starting any new medication and to make sure that this is the right choice for you. If you have any questions about metformin or if you’re thinking about starting taking it, be sure to talk to your doctor.
- The blog owner is not responsible for any adverse effects or consequences resulting from the use of any suggestions or information provided in this blog.Eden is not a medical provider.
- As stated, saxagliptin, when administered alone has a weight-neutral effect on T2DM patients’ weight, or may even produce weight loss when administered in conjunction with metformin
- In a small study, it was reported that the addition of metformin to an antagonist protocol improved the oocyte quality in PCOS patients undergoing IVF Doldi et al. 2006.
- Increasing water intake is an easy and effective way to support weight loss and overcome a weight loss plateau while taking Metformin.
- If you're diagnosed with acute cholecystitis, you'll probably need to be admitted to hospital for treatment.
- American Diabetes Association (ADA) 1997 diagnostic criteria (8) were used to establish the primary study outcome of diabetes defined by fasting plasma glucose ≥7.0 mmol/L (≥126 mg/dL) measured semiannually or 2-hour plasma glucose of ≥11.1 mmol/L (≥200 mg/dL) after a 75 g oral glucose load measured annually, with confirmation within six weeks.
- This enhances glucose uptake and utilisation by the body.
- This mindset could lead to increased food intake and subsequent weight gain, leading to misunderstanding the medication’s true effects.
However, it should be noted that the synergistic effect of metformin added on to other antidiabetic medications has not yet been reported. In the past decades, new classes of antidiabetic drugs have been approved to be used in T2DM patients. However, despite modest effects being observed, metformin monotherapy decreased liver transaminases and hepatic fat content. The effect of long-term treatment of metformin on liver-related adverse events are currently unclear, knowledge surrounding this is desirable. Metformin is taken up in the liver via organic cation transporter-1 (OCT1).54 Hepatic uptake of various drugs via this transporter has been shown to have differ between species, for example between mice and humans.55 To our knowledge, no previous research had explored the species difference of metformin uptake. These models generate uniform fatty rodents and the effect of metformin might be seen more clearly than in a human study in which the participants had various degrees of severity of fatty liver and concurrent metabolic derangements. Metformin treatment significantly decreased liver fibrosis evaluated by noninvasive measurement in NAFLD patients.46 However, inconsistent report exists. These patients’ characteristics suggest longer NAFLD disease duration and less insulin resistance in the enrolled patients. Metformin was shown to be beneficial in patients with NAFLD compared to baseline. One study included the patients who underwent liver biopsy and were diagnosed as NASH.51 The induction of autophagy enables cells to reutilize their own constituents for energy, one of the approaches for NAFLD treatment.41 The downregulation of SIRT1 expression and autophagy induction in the liver of ob/ob mice were restored following treatment with metformin.13 Additionally, metformin was shown to inhibit the STAT3 pathway,20 the pathway in which inhibition also induced autophagy.42 All these reports are summarized in Table 3. These findings provide a potential mechanism for metformin in treatment of NAFLD by alleviating inflammation in the liver and decreasing oxidative stress. While the markers of inflammation decreased, the oxidative stress parameters glutathione and superoxide dismutase (SOD),25 and the antioxidant protein peroxiredoxin 6 (PRDX-6)38 increased after metformin treatment. Proteins involved in mitochondrial lipid oxidation were up-regulated following metformin treatment,38 suggesting the potential effect of metformin in increasing mitochondrial lipid oxidation. The findings from in vitro studies demonstrated that metformin could reduce lipid accumulation13 and de novo fatty acid synthesis.14-16 Several proteins have been shown to be essential to the regulation of hepatic de novo lipogenesis. Relevant publications in the PubMed database were included in this review, the search terms used being “metformin” and “NAFLD,” “NASH” and “diabetes.” Only the articles published in English were reviewed. In addition, this review focuses on the efficacy of metformin as a monotherapy and as a combined therapy with other antidiabetic medications. Evidence has also suggested possible effects of metformin as regards the prevention of hepatocellular carcinoma tumorigenesis.
Although AMPK activation is a key target for metformin’s anti-tumour activity, there is evidence that metformin is also independent of AMPK. Due to its hydrophilic and cationic nature, metformin requires cation-selective transporters to enter cells and activate AMPK . One of the mechanisms of metformin’s anti-tumour action is the activation of adenosine monophosphate-activated protein kinase (AMPK), which is also involved in the anti-diabetic effect of metformin. It was shown that metformin is effective in inhibiting the proliferation and metastasis of breast cancer cells and inducing their apoptosis following multiple pathways 52, 53. However, the authors suggest that the published results should be interpreted with caution because retrospective data are at risk of bias . The authors suggest that more prospective studies are needed to elucidate the mechanisms and causality. Home use of metformin for more than 90 days during the year before hospital admission was an independent variable in the analysis. Patients included in the analysis were aged 18 years or older, had type 2 diabetes or obesity, and were admitted to the hospital for COVID-19, confirmed by PCR. In 2020 a meta-analysis evaluating the effect of metformin on the survival of women with ovarian cancer was published. Nonetheless there is insufficient evidence that metformin in combination with other agents that induce ovulation increases live birth rates . However, it has since been confirmed that metformin alone does not increase the rate of miscarriage when its use is discontinued early in pregnancy. Moreover, the use of metformin during pregnancy in women with polycystic ovary syndrome reduces the incidence of early pregnancy loss and preterm labour, and protects against foetal growth restriction .
In the present study, all models found time-dependent associations of compensatory insulin secretion and of weight change from baseline with the risk of developing diabetes. These results are consistent with previous DPP analyses showing that effects of the ILS and metformin interventions on reducing diabetes incidence were largely, but not entirely, attributed to weight loss (12, 13). For the same levels of weight loss, metformin decreased insulin secretory demand more than ILS (predicted distances along the baseline SMA line were smaller for metformin, Table S4 (16)). Beneficial weight loss effects in insulin secretion compensation were greater in the ILS intervention than in metformin (predicted distances away from the baseline SMA line at year 1 were greater for ILS, Table S4 (16)). This suggested that improvements in insulin sensitivity and beta-cell function were more prominent from baseline to year 1; however, as for weight loss, the effects of both ILS and metformin were attenuated after year 1. In both the TVCMs and the JMs, baseline weight and weight loss from baseline mediated over 49% of the effects of both treatments (53% for ILS and 50% metformin in the TVCM; 82% for ILS and 72% for metformin in the JM). Only the TVCM that did not adjust for baseline weight and weight change from baseline indicated that ILS significantly reduced diabetes risk compared with placebo (Tables 3 and 4). For the distances along the baseline line (insulin secretory demand), adding weight loss to the models attenuated the effects of ILS on them in all 3 years; this also happened for metformin but to a lesser extent. The treatment effects for both ILS and metformin were attenuated once we added weight loss to the models. In this study, we used repeated measurements of insulin secretion and sensitivity to investigate how abnormalities in both develop relative to each other and to determine whether treatment, time, weight change, or combinations of these variables affect their interaction during the progression from IGR to diabetes. ILS participants had the biggest movement to the right of the baseline curve after 1 year of treatment (11), and among them weight loss was the primary factor contributing to reduced risk of diabetes (12). By lowering blood glucose levels, metformin helps to regulate insulin levels and improve insulin sensitivity. Metformin is typically prescribed for people who are overweight or obese, as it can help improve insulin sensitivity and reduce the risk of developing type 2 diabetes. Studies have shown that people taking metformin for weight loss can expect to lose around 5-10% of their initial body weight over a period of 6-12 months. Metformin is primarily prescribed for people with type 2 diabetes to help manage their blood sugar levels. The combination of reduced insulin levels, increased fat breakdown, and suppressed appetite makes metformin an effective weight loss aid. This leads to a reduction in blood sugar levels, which in turn reduces the amount of insulin in the body. By understanding the underlying mechanisms, we can harness the potential of metformin as a weight loss tool, particularly for individuals with insulin resistance and type 2 diabetes. In fact, metformin has been shown to be effective in reducing body weight in individuals without diabetes, making it a potential treatment option for obesity. While we consider metformin to be a weight neutral diabetes medication, in clinical practice we see that some people do seem to lose weight when they start using it.

Originally prescribed for managing Type 2 diabetes, metformin has shown promising effects on cellular aging, inflammation reduction, and metabolic health. On the other hand, traditional metformin primarily works by reducing liver glucose production and may offer modest weight loss but not as significantly as Trijardy. One of the biggest advantages of Trijardy is its potential to support weight loss due to the action of empagliflozin, which helps the body excrete excess glucose. Unlike standalone metformin, Trijardy offers a multi-targeted approach to blood sugar management by addressing glucose control from different pathways. Since it is generally well tolerated, metformin for prediabetes is a valuable tool for long-term blood sugar regulation. Many women with PCOS struggle with weight gain and infertility, and metformin for PCOS helps improve insulin sensitivity, making weight loss more achievable. Those struggling with appetite or insulin imbalance may benefit from choosing metformin ER 500 mg as part of their metformin weight loss regimen. One notable trial found that participants taking metformin ER 500 mg lost more weight over a year compared to those on placebo. Additionally, the drug can suppress appetite in some users, contributing further to metformin weight loss benefits. Instead, metformin and weight loss is one of the reasons it's sometimes prescribed off-label for conditions like PCOS or prediabetes. However, clinical studies have consistently shown that metformin and weight loss often go hand in hand. Despite being a glucose-lowering drug, many still ask, does metformin cause weight gain? There are many FDA-approved weight loss drugs that patients seeking medical intervention can discuss with their doctor. Weight loss may be a potential secondary benefit of metformin use for type 2 diabetics who are overweight or obese, but using the drug as a standalone medication for this purpose seems illogical. There’s significantly more research backing metformin’s efficacy for diabetes than for weight loss. However the weight loss effect appears moderate, and the drug is not approved by the FDA to treat obesity; only to treat type 2 diabetes.
Learn about causes, diagnosis, treatment options, and when to seek medical help. Discover common causes, symptoms, treatment options, and when to seek care for lasting relief. Learn how UTIs in men can affect men’s sexual health and sometimes lead to erectile dysfunction (ED). Using three medications together is a regimen also known as triple therapy. Wellbutrin (bupropion) is a generic medication typically prescribed to treat depression. Weight loss is a common side effect of the drug, but it’s not its main purpose. Due to high demand, the availability of metformin is currently limited. Metformin is one of the more affordable diabetic medications. Studies show it is possible for men to experience erectile dysfunction after starting metformin, but it’s rare. While taking metformin, it’s important to stay hydrated. It can minimize the risk of liver damage and liver fatty disease. Metformin starts working in the body as early as three hours after taking a tablet. At most, a healthcare provider may recommend up to 2,000 mg daily. A healthcare provider may increase your dosage in 500 mg increments to help prevent gastrointestinal symptoms. It’s not uncommon for a healthcare provider to recommend increasing your dosage over time. The type of tablet prescribed to you determines how often the medication is taken. Recommended metformin dosages can range from 500 to 2,550 milligrams (mg). Be sure to speak with a healthcare provider about the proper dosage for you. Metformin is an oral medication that should be taken with food. Weight gain is a common side effect of antipsychotic drugs like olanzapine (Zyprexa) and clozapine (Clozaril).
Too much insulin can also stimulate the ovaries to make more androgen. It can result in excess facial and body hair, acne, male-pattern baldness, and other signs of hyperandrogenism. PCOS can make the body produce too much of a hormone called androgen, usually present in small amounts in women. These hormonal imbalances can contribute to weight gain, especially in the abdominal area. This hormonal imbalance affects the ovaries and can lead to complications such as irregular or skipped periods, excess body hair, acne, pelvic pain, difficulty getting pregnant, and other symptoms. Polycystic Ovary Syndrome (PCOS) affects millions of women worldwide, causing hormonal imbalances that lead to weight gain, irregular periods, and even difficulty conceiving. Are you tired of struggling with weight gain and the frustrating symptoms of PCOS? Nimisha Gupta is a content writer at Fitelo, a health and wellness platform. Metformin appears to have a protective effect on the development and progression of cancer, although the results of other studies do not confirm this relationship. This is important because metformin is well tolerated and can effectively reduce the dose of cytotoxic chemotherapeutic agents in combination therapies without reducing their anti-cancer efficacy. Moreover, the authors found that the combination of metformin and propranolol reduced the metastasis and proliferation of 5-FU-resistant colon cells . Metformin in combination with aspirin significantly inhibited the growth of pancreatic cancer cells in both in vitro and in vivo studies. In another study in a mouse model, animals with transplanted breast cancer tumours received in their drinking water i.a. This is important because metformin is well tolerated and can practically lower the doses of cytotoxic chemotherapeutic agents in combination therapies without losing efficacy . A significant benefit for overall survival was observed in patients with stage II and III disease . Administration of metformin increases GLP-1 concentration and the bile acid pool in the intestine, and alters the microbiome . It was shown that the use of metformin after diagnosis (1 year of use) resulted in a significant reduction in mortality. Metformin in combined with another drug increases the effectiveness of chemotherapeutic agents and radiotherapy.
How much weight can you lose on metformin vs. Ozempic?
Because the largest difference in body weight was in the ethnic Chinese population we conducted a sensitivity analysis by excluding these studies (Fig. 7). Subgroup analysis shows that the five trials which included first episode patients −5.94 kg (95 % CI 6.75 to −5.12) showed a much larger difference in mean body weight change than trials of chronic patients −2.06 kg (95 % CI −2.71 to −1.41) (Fig. 6). Only one trial reported diarrhoea was significantly more in the metformin group compared to placebo . De Silva et al. reported one discontinuation due to dizziness in metformin group and 3 due to development of diabetes in placebo group . Ten studies of adults were included and two studies found a significant difference between metformin and placebo. Forest plot of mean change in BMI in patients treated with metformin versus placebo Of them seven studies in adults and one in children showed that there was significant difference in BMI between metformin and placebo. Five studies reported a significant difference between metformin and placebo. Six studies reported the change in body weight percentage 16, 17, 25–28. Secondary outcome measures were change in BMI (kg/m2), fasting blood sugar (mg/dl) and insulin resistance index (IRI). The primary outcome measure of efficacy was mean change in weight in kg between pre-treatment and end of study weight.
It lowers testosterone and improves heart health. Studies show it helps with weight, hormones, and metabolism. Metformin is a popular choice for treating PCOS, especially for overweight women. It fights insulin resistance, a common issue in PCOS. Metformin works by stopping the liver from making too much glucose. Metformin plays a key role in managing PCOS by targeting insulin resistance. About 50% to 70% of women with PCOS have insulin resistance. In some studies, 53.7% of young unmarried female students showed PCOS symptoms. These symptoms can greatly affect daily life and may lead to other health problems. This will help us understand if it can cause weight gain. We need to look into how metformin affects women with PCOS. For women with PCOS, managing symptoms and weight is hard. A different medication or different combination of medications may be a better solution for you. A medication that works for one person may not work the same way for another. The excess stored energy will be changed to fat if energy expenditure remains low, and as a result, weight increases. Weight and diabetes, especially type 2 diabetes, are closely linked. HIt and HIs contributed to the design of the study, analysis and interpretation of data, and drafting of the manuscript. The results of this study have several limitations that must be considered.
Despite guidelines commonly recommending a 500 mg starting dose, many clinicians now advocate for more gradual titration in patients with known sensitivity. Initiating metformin at a very low dose—sometimes as little as 125 or 250 mg per day—and increasing slowly over weeks is often enough to minimize early intolerance. These bacteria can support glucose regulation, but also promote fermentation, gas, and shifts in hormone production. Equally important is the way metformin changes the gut microbiota. In diabetic mice, metformin inhibits NHE3 via AMPK activation, leading to increased fecal water content (Han & Yun, 2022). A major contributor is the drug’s ability to increase intestinal transit. The net result, in some patients, is loose stools that range from occasional discomfort to severe, daily disruption. For others, it’s a persistent disruption that forces reevaluation of the medication altogether. While often brushed off as a temporary inconvenience, metformin-induced diarrhea can significantly impact adherence and quality of life. Metformin has long been the foundational therapy for type 2 diabetes, recognized for its efficacy, cardiovascular protection, affordability, and favorable safety profile. Metformin also has AMPK-independent effects on the liver that may include inhibition of fructose-1,6-bisphosphatase by AMP. Metformin is a complex drug with multiple sites of action and multiple molecular mechanisms. It is possible that recent observations, consistent with the ability of metformin to prolong mammalian lifespan 54, 55, may, at least in part, be due to suppression of this cytokine. Interestingly, one of the cytokines suppressed by metformin is C-C motif chemokine 11 (CCL11), which has previously been found to contribute to age-related cellular and tissue dysfunction. Metformin has, for example, been shown to have direct effects on inflammation, including effects on NF-κB signalling and differentiation of monocytes into macrophages 52, 53, as well as suppressing proinflammatory cytokines from these macrophages. Were the most consistently observed effects on the microbiome across datasets from different countries . In animal studies, which was linked to reduced adipose tissue inflammation and suppressed postprandial hyperglycaemia . Consistent with this interesting concept of metformin’s ability to affect host metabolism indirectly, metformin expanded the gut population of Akkermansia spp. A third potential mechanism of intolerance may be due to the impact of metformin on the intestinal microbiome (see later).
Obesity is one of the most common health conditions in the United States. Consulting a healthcare professional is advised to create a personalized plan that aligns with individual health goals and needs. In rare cases, Metformin can lead to a serious condition known lactic acidosis, particularly in individuals with impaired kidney function or other risk factors. Common gastrointestinal issues include nausea, diarrhea, and stomach upset, especially when starting the medication. While Metformin is generally well-tolerated, it can cause side effects in some individuals. However, consult with a healthcare provider before using these, as safety and effectiveness can vary widely. They can determine if the medication is appropriate and safe for you, particularly if you have pre-existing conditions. Though rare, metformin can cause lactic acidosis, a serious condition that demands immediate medical attention. Our comments section is a place where readers can engage in healthy, productive, lively, and respectful discussions. “Walking is one of the best exercises you can do while taking metformin,” Dr. Pobee says. But researchers continued to track participants for 15 years and found that they also kept the weight off, and even continued to lose. Herein, we discuss the potential mechanisms by which metformin decreases appetite and opposes unfavorable fat storage in peripheral tissues. We demonstrated in vivo that the administration of metformin may cause visceral fat reduction through a possible mechanism of fat oxidation enhancement that is independent of its appetite suppressive effect. In conclusion, it is feasible that the long-term administration of metformin brought about a shift of the fuel source for fat oxidation in both human and animal experiments, and a significant decrease of visceral fat volume was noted in animal experiments. While this dose is similar or somewhat smaller than the dose of previous reports in which metformin was administered to rats through their drinking water 35, 36, it is estimated to be approximately 10 times higher than that in human subjects (about 30mg/kg per day). Finally, the dose of metformin that was used for rats was around 250mg/kg per day, with water consumption taken into consideration. Second, because it is complicated and labor intensive to directly measure insulin sensitivity, we instead measured the serum insulin concentrations to estimate insulin sensitivity. Metformin’s effects on fat oxidation coincide naturally with its activation of AMPK in the liver, and the inactivation of ACC by AMPK results in a decrease in malonyl-CoA, higher CPT-1 activity and the enhancement of fatty acid oxidation . In contrast, the previous studies measured the average RQ for a whole day.
You can lose weight without exercise, but it's harder to do. To lose weight, you need to lower the total calories you take in from food and drinks. You set action goals so that you can make healthy changes. You can list a healthy outcome that you aim to have. Those conditions include heart disease and type 2 diabetes. Losing 5% of your current weight may be a good goal to start with. If you prefer to keep your weight-loss efforts private, take some steps to stay on course. Think of your goals on days when you don't feel like eating healthy foods or moving more. Maybe you want to boost your health or get in shape for a vacation. What will give you the burning desire to stick to your weight-loss plan? No one else can make you lose weight. Lowering stress can help you make long-term healthy lifestyle changes. Talk with your healthcare professional if you need help taking charge of stress.
The results of this study were published in the Journal of Diabetes Research. This means you’ll lose weight faster than if you were just trying to diet on your own! It helps control appetite by increasing insulin sensitivity so that you’re less hungry throughout the day and can eat less food without feeling deprived or hungry. So if you’re trying to lose weight but not having much success reaching your goals so far…we recommend starting with Metformin first! Well, when you have more abdominal fat than other types of fat on your body (such as thigh or hip), it’s an indication that there are metabolic issues at play. It’s a bit of a cliché, but the truth is that no one ever said losing weight would be easy. This is much better than what I was seeing at first, so now I’m confident that this method will help me reach my goal weight by the end of this year. Metformin side effects are generally mild and include nausea, vomiting, diarrhea, heartburn and abdominal pain. It works by decreasing the amount of sugar produced by the liver and increasing the sensitivity of cells to insulin. It’s also been found to protect against age-related cognitive decline, diabetes complications such as heart disease and eye disease, and other conditions like polycystic ovary syndrome (PCOS). The participants were divided into two groups, one receiving Metformin and the other receiving a placebo. A total of 108 obese individuals with a BMI greater than 30 were included in the study. There are plenty of ways for you to improve your health without losing any more pounds than necessary–and those methods won’t come at the cost of feeling sick every day either! It’s important to note that metformin is not a magic pill; it won’t magically melt away pounds without any effort on your part! I’ve been taking metformin for two months now, and I’m happy to report that it’s working! Focus on eating a variety of nutrient-dense foods, including fruits, vegetables, whole grains, lean proteins, and healthy fats. When taking Metformin, it is important to be mindful of your diet and exercise habits to prevent weight gain. When taking Metformin, it’s important to be aware of its impact on weight management. Metformin is generally well-tolerated but can cause side effects such as gastrointestinal upset, including diarrhea and nausea.
- Women with PCOS, a condition often accompanied by insulin resistance and weight gain, may also find metformin beneficial.
- Metformin 850 mg is a higher dosage often prescribed when lower doses fail to achieve the desired blood sugar control.
- Keep a fast-acting glucose source nearby and consult your doctor if symptoms occur frequently.
- Acute cholecystitis is potentially serious because of the risk of complications.
- The efficacy of liraglutide (1.2 mg, 1.8 mg, 2.4 mg, or 3.0 mg) was assessed in obese nondiabetic individuals in a 20-week double-blind, placebo-controlled trial with an open-label orlistat comparator .
- It’s not uncommon for physicians to recommend metformin as a weight management aid even though the FDA has not approved metformin as a weight loss medication.
Instead, it’s essential to work with your healthcare provider to gradually taper off the medication under close medical supervision. It’s important to note that metformin is not a substitute for a healthy diet and regular exercise. The most common side effects of metformin include gastrointestinal symptoms such as diarrhea, nausea, and stomach cramps. To achieve significant weight loss, you will need to make sustainable lifestyle changes that you can maintain in the long term. The amount of weight you can expect to lose while taking metformin will vary depending on a number of factors, including your starting weight, diet, and level of physical activity. In fact, one study found that individuals who took metformin in combination with a low-calorie diet and regular exercise lost an average of 10 kg (22 lbs) over a period of 12 months. This leads to a decrease in the amount of glucose available for energy, and as a result, the body begins to break down stored fat for energy instead. Metformin is an oral anti-diabetic medication that is commonly used to treat type 2 diabetes. It’s essential to remember that metformin is most effective when combined with healthy lifestyle modifications. While individual results may vary, a typical weight loss range of 1-5 kg (2.2-11 lbs) over 6-12 months can be expected. John, a 35-year-old obese individual without diabetes, started taking metformin (2,000 mg/day) and adopted a more aggressive lifestyle modification plan. Sarah, a 45-year-old woman with type 2 diabetes, began taking metformin (1,000 mg/day) and made moderate lifestyle changes. Metformin increases lipolysis, resulting in increased fat burning and weight loss. Metformin increases insulin sensitivity, allowing glucose to enter cells more efficiently and reducing glucose production in the liver. Start your weight loss journey with a friend and send them $50 off weight loss - on us. We’ll also discuss if it's safe to mix them, and other risks and side effects of drinking as a type 2 diabetic. Are they the game-changers they're touted to be in the realm of weight loss? It is also gaining attention for its off-label use, helping patients lose weight.
Delhi-based Megha Hooda was facing weight gain issues due to her PCOS. Generally, Metformin is used to treat Type 2 diabetes, but it is also helping women with PCOS shed those stubborn pounds. PCOS, or Polycystic Ovary Syndrome, affects millions of women worldwide through hormonal imbalances that can lead to weight gain and difficulty losing weight. Don’t forget the importance of contacting a registered dietitian or healthcare professional for expert advice on your diet. It includes both prescription and over-the-counter medications. Drink plenty of water while taking Metformin to stay hydrated and support overall health. Common side effects may include gastrointestinal issues such as nausea, diarrhea, or stomach discomfort. They may recommend blood tests or other assessments to evaluate your response to Metformin. It can help provide sustained energy and support overall health. Focus on a well-balanced diet with adequate lean protein, healthy fats, fiber-rich foods, and complex carbohydrates. Take Metformin exactly as prescribed by your healthcare professional. Also, you must know that everyone reacts differently to medications. Now how much weight can you lose with Metformin for PCOS? Thus, Metformin may be the solution for how to lose weight with PCOS by addressing this underlying hormone imbalance, as androgens lead to weight gain, particularly in the abdominal area. High androgen levels are often responsible for symptoms such as excessive hair growth and acne. It helps regulate glucose metabolism and decrease appetite while increasing feelings of fullness after meals. It can lead to decreased hunger, reduced food intake, and improved weight management. This hormonal imbalance contributes to weight gain and difficulty losing excess pounds.
A six-month, randomized, double-blind, placebo-controlled research study was conducted in 2022, employing 156 obese subjects to examine the impact of this unique combination on weight loss . The patients who received acarbose and had significant variations in their waist-to-height ratios experienced average body weight losses of 3.63 kg (5.28%) . This benefit is contingent on baseline body weight, though, since patients with higher baseline body weights and/or BMIs experienced greater weight loss . In obese people with a BMI greater than 35 kg/m2, the experiment found that metformin treatment significantly reduced BMI as compared to baseline . A total of 85 T2DM patients with non-alcoholic fatty liver disease participated in a 24-week randomized experiment to evaluate the effects of gliclazide, liraglutide, and metformin on body composition . Moreover, 56% of patients in the metformin group kept their weight loss at least at 5%, as opposed to 43% of patients in the lifestyle group . Moreover, the only biguanide medication that has received FDA approval for the treatment of hyperglycemia in T2DM patients is metformin . Current research has demonstrated that metformin offers additional therapeutic advantages than lowering glycemia, such as extending life, reducing body weight, and decreasing the risk of developing cancer . Several studies have demonstrated that some antihyperglycemic medications can help T2DM patients lose weight, whereas other antihyperglycemic medications are weight-neutral . Liraglutide and semaglutide are two of the antihyperglycemic medications that are utilized and have FDA approval for the treatment of people with type 2 diabetes 13,14.
However, hypertrophic and functional improvements following RT vary among individuals, with some people completely failing to experience muscle hypertrophy (Stec et al., 2017). Accordingly, RT has also been shown to improve activities of daily living, overall health, and quality of life in elderly individuals (Hunter, McCarthy, & Bamman, 2004). Analyses of vastus lateralis muscle biopsies showed that metformin did not affect fiber hypertrophy, or increases in satellite cell or macrophage abundance with PRT. There was a trend for blunted strength gains in metformin that did not reach statistical significance. Suppression of androstenedione was significant with both metformin doses, but there was no clear dose relationship. No comparative examination of weight changes or metabolite responses to different doses has been reported. Similarly, hunger ratings were significantly lowered after metformin, and the effect was most pronounced after the administration of 1700 mg of metformin. The 1700-mg metformin dose had the most marked appetite suppressant action. Additionally, future research should strive to elucidate if there is a relationship between the dosage of metformin and exercise intensity and/or type and determine if increases in metformin dosage need to be congruent with increases of exercise intensity. Understanding the relationship between the timing of metformin and exercise will help guide future prescriptions and help mitigate the less favorable outcomes that are observed when these two modalities are combined. While these treatments have similar targets through which they act, in recent years, research has shown the lack of synergistic effect of the two. Lifestyle, lack of exercise, poor diet were shown to lead to the dysregulation of glucose homeostasis and the development of IR. Potentially, understanding of metformin dose and exercise intensity, as well as the energy expenditure threshold, could contribute to the development of a dose/response model, allowing for better adjustment of the prescription. Potential interference between exercise modalities could be at the level of the mitochondrial electron transport chain, where exercise-induced energy demand is not met due to the complex 1 inhibition by metformin, resulting in a lower ATP output. The possible disparity in the effects could be attributed to the exercise intensity difference, where Ortega et al18,19 used 4 bouts of 4 minutes at 90% of maximal heart rate (HRmax) interspersed by 3 minutes of cycling at 70% HRmax, while Myette-Côté et al17 implemented a moderate-intensity walking at 85% ventilatory threshold for 50 minutes. Takao et al74 demonstrated the direct relationship between increased postprandial hyperglycemia and risk for the development of CVD. Considering that both groups had a similar improvement from the baseline to the post-exercise regimen, it can be rationalized that improvements in glycemic control can be achieved with exercise alone, regardless of metformin therapy.
After starting metformin, John noticed an improvement in his blood sugar levels and a gradual decrease in his weight. Achieve your weight loss goals with Semaglutide—a powerful GLP-1 medication that reduces hunger and helps regulate blood sugar. Achieve your weight loss goals with Tirzepatide—a dual-action medication that curbs appetite and regulates blood sugar. This could be a sign of another health problem.The effect of metformin on blood sugar means people should keep an eye on their symptoms. Metformin may also positively influence patients overall cardiometabolic risk profile, including improved blood glucose control.12 The GDG issued a strong recommendation in support of metformin as an alternative first-line strategy where diet and lifestyle interventions are ineffective, inappropriate or unacceptable. Member experience was that patients typically place a greater value on managing AIWG with metformin, compared with limiting their tablet burden and risk of transient gastrointestinal side effects. The treatment of diabetes should not only focus on glycemic control as its sole intention, but it should factor in the effect of these various agents on weight as well, since obesity aggravates insulin resistance, beta cell failure, and cardiovascular risk. Subjects who received combination treatment achieved greater weight loss (−11.3 kg) compared with pramlintide alone (−3.6 kg) or placebo (−2.1 kg). Patients treated with orlistat had a statistically significant greater decrease in body weight (−3.8 kg) than placebo-treated patients (−1.4 kg) and a larger decrease in HbA1c compared with placebo (−0.74% versus −0.31%). More patients in the orlistat group achieved a weight loss of ≥10% compared to the placebo group (26% versus 15.6%). In a double-blind, open-label study, metformin-glibenclamide combination was compared with metformin-rosiglitazone in 318 type 2 diabetes patients uncontrolled on metformin monotherapy (mean HbA1c ~8.5%) . Patients in the metformin group had a greater weight loss of 2.1 kg compared to placebo but less compared with the lifestyle-intervention group of 5.6 kg. This study was conducted to evaluate metformin in the prevention or delaying of the progression of type 2 diabetes in high-risk patients . Further studies examining the effects of antiobesity drugs and GLP-1 agonists to combat insulin-induced weight gain are needed. Thus, the effect of antidiabetes agents on weight and obesity status is an important consideration when treating patients with diabetes since obesity, itself, aggravates insulin resistance and cardiovascular risk.
Serious side effects are rare and happen in less than 1 in 10,000 people. You may also need to eat a starchy carbohydrate, like a sandwich or a biscuit, to maintain your blood sugar for longer. It's also possible for your blood sugar to go too low while you're asleep. Metformin does not usually cause low blood sugar (known as hypoglycaemia, or "hypos") when taken on its own. Like all medicines, metformin can cause side effects, although not everyone gets them. Although in-person care in an inpatient or outpatient treatment setting can be helpful for many, more and more people are considering virtual care, given its flexibility and accessibility. Weight stigma and fatphobia are even present in healthcare, including diagnostic and treatment settings. In one case study, a woman with anorexia-binge purge type misused metformin, which she’d bought on the black market online. Furthermore, overdosing on metformin can also impair kidney function due to poor blood flow.2 Serious metformin risks may include chest pain or a rash, which could indicate an allergic reaction.1 If you experience a dangerous adverse effect, call 911 or your doctor immediately. Metformin increases the feeling of fullness and helps people lose weight by reducing the amount of food eaten throughout the day. For additional information on metformin, please talk to your health care provider. Systematic review of the benefits and risks of metformin in treating obesity in children aged 18 years and younger. Advances in the mechanism of metformin with wide-ranging effects on regulation of the intestinal microbiota. Talk with your clinician to see if metformin aligns with your health needs and long-term goals.
We also aimed to assess the effect of this combined treatment on waist circumference, triglycerides, blood pressure, and inflammatory cytokines. We suggest to compare the consequences of metformin withdrawal after long-term therapy as opposed to the consequences of metformin withdrawal immediately after the maximum treatment effect is achieved, usually after the first year. The protective effect of the long-term use of metformin in reducing the risk of unopposed endometrial proliferation and endometrium cancer should also be evaluated. The high rate of drop outs not related to intolerance and side effects could be decreased in clinical practice by discussing the realistic treatment goals and potential benefits of long-term intervention. The overweight-obese and oligomenorrheic women should be prioritized treatment candidates. We suggest that treatment decisions for metformin are based on BMI, oligomenorrhea and biochemical hyperandrogenism regardless of the glycemic status. Another point of concern is the high rate of drop-outs and the effects that may have on the study outcome. There have been very limited studies of the natural history of glucose homeostasis in this population. Our patients in whom menstrual regularity improved after first year had less frequent bleedings at baseline when compared with those with no improvement in menstrual frequencies. Given that PCOS is characterized by progressive weight gain from menarche (25, 26, 27), the difficulty in stabilization of BM is expected to be even more pronounced in PCOS when compared to general population. Notably, after 2 years, remarkable drop out that had not been related to metformin intolerance or side effects was seen in this real-life setting. Those with higher baseline BM, BMI, less frequent menstrual bleedings and higher levels of androgens were identified as subgroups where metformin’s effect seemed to be enhanced. Development of mild anemia with normal mean corpuscular volume (MCV) values were documented in five patients, all had normal levels of vitamin B12. As seen from the patient’s perspective, maximal treatment response achieved after first year was followed by a steady state and then the benefits of the steady state and other potential beneficial effects of long-term treatment were not sufficiently discussed with them. Mean levels of fasting glucose did not change significantly after first year follow-up and not during the following years. Twenty-five patients gained weight after 1 year, 60% of those who had 11 or 12 menstrual bleedings at baseline. BMI, body mass index; DHEAS, DHEA sulphate; FBG, fasting plasma glucose; FSH, follicle-stimulating hormone; LH, luteinizing hormone; MF, menstrual frequencies; s.d., standard deviation. Glucose levels were determined using a standard glucose oxidase method (Beckman Coulter Glucose Analyzer, Beckman Coulter Inc CA, USA).
- If your blood sugar levels are stable, taking metformin should not affect your ability to drive, cycle or use machinery and tools.
- Clinical studies of overweight women with PCOS have found that endocrine disorders can lead to infertility .
- When taking Metformin, it is important to be mindful of your diet and exercise habits to prevent weight gain.
- However, the extent of weight loss can vary among individuals, and factors such as diet, exercise, and overall health play a significant role in the outcome.
- If your blood sugar levels become too low, this can reduce your concentration.
- The diagnosis of PCOS was established using the Rotterdam criteria with all included patients characterized as phenotype A.
- Furthermore, metformin also promotes the expression of the tight-junction protein occludin113,124–126, which reduces the translocation of lipopolysaccharide, colonic inflammation and dysfunctional gut permeability.
- Is used for the treatment of acromegaly (caused by hypersecretion of growth hormone).
- The mechanism of weight reduction, by increasing urinary excretion of glucose, has created the perception that the weight reduction may be via water loss as well as lost calories, but studies have shown reductions in both visceral and subcutaneous adipose tissue 74,75.
- Using this medicine with any of the following medicines may cause an increased risk of certain side effects, but using both drugs may be the best treatment for you.
Different treatment options, doses, duration, and enrolment of different populations may have led to obvious heterogeneity, and we need to interpret the results carefully. We have to admit that this study may have some serious limitations. In the future, there is a need for a greater diversity of research, such as cooperation between multiple centres, more rigorous clinical reports, and prospective studies. In both studies, different laboratory tests were used, which may have an impact on the comprehensive measurement results. We performed a sensitivity analysis of the included RCTs and found that two studies may be a source of most of the heterogeneity. Second, the study distribution between the twelve RCTs was included, which may affect the meta-analysis results. Insulin resistance will cause hyperinsulinemia, which directly affects the role of ovarian receptors, inhibits insulin-binding protein and sex hormone-binding protein, while freeing testosterone and increasing ovarian androgens. The production of polycystic ovary syndrome is directly related to the abnormality of insulin. The antireproductive effect of metformin helps correct this phenomenon . It can be considered as abnormal gonadotrophic secretion in women with overweight PCOS, which makes FSH in an abnormal secretion stage . Obese women with PCOS show lower ISOGTT and higher LH to stimulate androgen secretion, triggering insulin resistance and excessive androgens .
The study of Lundby-Christensen found a decrease of 2.6 kilograms in the metformin group after 18 months. The forest plot clearly shows that the length of the intervention increases the body weight change. Three studies applied the “Last observation carried forward” method 23, 24, 28, one study imputed missing data by application of the multiple imputation method , one study used the summary mean of nonmissing values over the entire observation period , whereas Robinson et al provided no information on attrition. In the study of Hermann et al , six different doses of metformin and glyburide were used, therefore outcome data and outcome recordings were not unequivocal, since they reported the pooled results of three dose combinations. Four of the studies received maximal score, and no study was considered low quality. Significant differences between the mean age of the placebo and metformin groups before randomization were indicated in two of the articles. With regard to the presence of small study effect, due to the low number of the included studies, the results of the Egger’s test are not informative; therefore they were omitted from the results. The differences were calculated by subtracting paired change after metformin from paired change after placebo. Therefore, the detailed assessment of the side effects of metformin in the elderly is of extremely high importance. Metformin seems to be a promising drug for aging prevention in humans , since in addition to its anti-diabetic actions, it also exerts anti-tumor and anti-aging effects . Metformin is also recommended as a combination therapy for patients with Type 2 diabetes .
There was a greater frequency of patients without any antidiabetic agents prior to metformin administration in the obese group (31%) than in the non-obese group (16%). The odds ratio (OR) and respective 95% confidence interval (95% CI) were determined to examine the strength of the relationship between the requirement of additional glucose-lowering agents (OHAs or insulin) and the clinical parameters based on a multiple logistic regression analysis. The triglyceride concentrations were not investigated in this study because fasting blood samples could not always be obtained for measurements. The clinical parameters, including BMI, HbA1c level and the history of medication were investigated from the baseline (the initial date of metformin therapy) for 3 years. It is similar in Japanese diabetic patients, for example, the mean BMI was 22.9 kg/m2 in men and 23.4 kg/m2 in women in the Japan Diabetes Complications Study (JDCS), which was a nationwide multi-center prospective study of type 2 diabetic patients . This effect appears to be maintained even after the observation period of this study, because metformin was limited to a relatively low dose in the non-obese group and the observed worsening in glycemic control over time can probably be attenuated by increasing the dose of metformin. HbA1c levels were significantly decreased compared with those at the baseline time. Current data indicates that up to 1 in 14 patients taking metformin for a period of 4 years develops Vitamin B12 deficiency and it is more likely common with high doses and longer use. Worse yet, metformin has been linked to an increased risk of developing dementia and Parkinson's Disease. In fact, there have been studies that suggest a nearly two fold increase in the likelihood of mental impairment in those taking metformin versus those who are not. Furthermore, studies in mice have shown that metformin promoted neurogenesis (i.e. generation of new neurons) and enhanced memory in rodents, as well as a overall reduction in oxidative stress. In Vitro studies (i.e. studies conducted in the lab in a non-biological context) showed that metformin could prevent the formation of amyloid beta peptides, one of the hallmarks of neurodegeneration in Parkinson's Disease. Interestingly, early studies indicated that metformin showed potential promise in treating and preventing certain neurological conditions such as Alzheimer's Disease and Parkinson's Disease. Current evidence suggests that metformin does in fact have a negative effect on memory, although more studies are needed. In this article, our pharmacist discusses whether or not metformin causes memory loss and how to reverse the effects. Feeling weak, dizzy, or fatigued may indicate low blood sugar.
However, when given as combination therapy, the opposing actions of these two drugs were cancelled out, leading to no change in lipolysis in scWAT. For example, we found that in scWAT, Mir alone increased lipolysis,29, 40 whereas Met suppressed it,32 consistent with previous studies. Interestingly, however, differential effects of Met, Mir, and Met/Mir on lipolysis in other adipose depots were identified. This is the case in our prevention model where increased BAT lipolysis by Met/Mir treatment was coupled with increased BAT thermogenesis. We are the first to report this depot‐specific expression profile of lipolysis in response to Met and/or Mir treatments under two distinct clinical contexts. Of particular importance, an additive augmentation of BAT thermogenesis was produced by Met/Mir in the treatment model, which could be responsible for the greater level of overall EE. This is the first evidence of how Met/Mir therapy produces additive properties to change EI and EE that neither drug could achieve alone. (B) In the treatment model, a diet‐induced obesity (DIO) phenotype was first established followed by 5 weeks of therapeutic treatments with Met and/or Mir. Moreover, we reported fat depot‐specific responses to Met/Mir treatment in thermogenesis, lipolysis, and fatty acid oxidation. These effects on BW were mainly achieved by activating BAT thermogenesis and promoting WAT browning, which led to enhanced EE, as depicted in the schematic diagram in Figure 9. Additionally, the weights of the liver, heart, lung, spleen, and kidneys in DIO mice remained unchanged (Figure S5M,N). Only Met caused a significant increase in the double product, while no change was observed after Mir or Met/Mir treatment (Figure S5L). Heart rate, MBP, and DBP (Figure S5H–J) were not affected by any treatment.
Talk to them if you notice any changes in your hormones or if you have any fertility worries while taking metformin. Metformin is mostly used to help with insulin resistance. At Healthon, we are here 24/7 to help you in this journey to good health. Doing regular physical activity and following a balanced meal plan can make metformin work better. Regular checkups and care help people use it in the best way. To learn more about why men and women want to lose weight, you can check out this article. This helps you be more safe if you are using metformin. Reading this guide will help you know more and make good choices for your health. This is really helpful for people dealing with conditions like polycystic ovary syndrome. When you understand HRT, you can also talk better about how metformin works. Still, if you see your healthcare provider for check-ups, these problems do not happen often. So, it is important to watch for these side effects.
So, does Metformin cause weight loss in people who do not have diabetes? Many people with PCOS struggle with obesity, and since Metformin improves insulin sensitivity, it can help regulate metabolism and possibly support weight loss. This makes it less effective as a weight loss drug, especially for people looking for significant results. While some studies suggest it can lead to modest weight loss, it is not classified as a weight loss drug. When insulin resistance occurs, blood sugar stays high, and the body produces even more insulin to compensate. Metformin’s primary function is to lower blood sugar levels by improving insulin sensitivity. Metformin can lead to gradual and modest weight loss in some individuals, particularly those with insulin resistance or type 2 diabetes. On the other hand, people without insulin resistance or those who do not adjust their diet or physical activity may not experience noticeable weight loss. While Metformin is not a weight loss drug, some people experience gradual weight reduction while taking it. Metformin is a widely used medication that lowers blood sugar, improves insulin sensitivity, and reduces sugar production in the liver. Some people also wonder, does Metformin cause weight loss, especially since weight loss is sometimes observed as a side effect of the drug. It belongs to a class of drugs called biguanides, which work by helping the body manage blood sugar more effectively. By the end, readers will have a clear understanding of whether Metformin is a reliable weight loss tool or if its main role is in managing blood sugar. Some people are also concerned about side effects and whether those side effects might contribute to weight loss. However, researchers and patients alike have noticed that in addition to lowering blood sugar, Metformin may also have an impact on weight. Instead, some studies show that people taking Metformin experience modest weight loss. Some people assume that any medication for diabetes will lead to weight gain, but Metformin is different. With the right approach, metformin can be part of a successful weight loss journey, helping you achieve your health goals safely and effectively. However, metformin is not a weight loss drug and should not be taken for this purpose without a doctor’s guidance.